Original Article
, Volume: 12( 12)Neurotoxicity of Acetamiprid in Male Albino Rats and the Opposite Effect of Quercetin
- *Correspondence:
- Rachid R , Department of Applied Biology, Tebessa University, 12000, Tebessa, Algeria, Tel: +213661259899; E-mail: r_rouabhi@yahoo.fr
Received: September 17, 2016; Accepted: November 15, 2016; Published: November 26, 2016
Citation: Gasmi S, Rouabhi R, Kebieche M, et al. Neurotoxicity of Acetamiprid in Male Albino Rats and the Opposite Effect of Quercetin. Biotechnol Ind J. 2016;12(7):113.
Abstract
The main aim of this work is to study the opposite effects of Quercetin against neurotoxicity induced by an environmental pollutant in albino rats; after administration of Acetamiprid at (3.14 mg/kg/day) of body weight orally during 90 days we have found that Acetamiprid has caused a neurotoxic effect (a decrease in relative brain weight, a decrease in lipids (0.54 ± 0.01), an increase of carbohydrates (1.43 ± 0.05) and total proteins (6.97 ± 0.02) of the brain), Acetamiprid has also an overall pro-oxidant effect, this is revealed by the significant reduction in the level of reduced Glutathione (5.01 ± 0.31), and activity of Glutathione peroxidase (0.042 ± 0.002) and catalase (1.26 ± 0.04) in the brain and on the other hand, we recorded an increase in enzyme activity of glutathione sâ€ÂÂÂÂtransferase (0.018 ± 0.002), the rate of MDA (0.08 ± 0.009) and Adversely affect the behavior and lifestyle. Our results have showed that supplementation of the Quercetin at (10 mg/kg of body weight/day) by oral way in rats treated with Acetamiprid, improved some biochemical parameters, and declined other adverse effects of Acetamiprid, and that the Quercetin appears to be a simple and effective antioxidant to reduce the imbalance between the formation of free radicals and antioxidant systems of the body and consequently reduced the intensity of oxidative stress induced by Acetamiprid.
Keywords
Quercetin; Neurotoxicity; Acetamiprid pollutant; Albino rats; GSH; GST; CAT; Brain
Introduction
After the second world war, pesticides have allowed the development of agriculture and contributed to the increasing and regulation of the agricultural production. The use of plant protection products has also limited or eradicated the number of very deadly parasitic diseases [1]. However, today, pesticides are believed to pose a risk for human health and the environment. They are indeed frequently implicated in the degradation of the quality of fresh groundwater and coastal waters, in the reduction of terrestrial biodiversity, found in agricultural areas and in natural contaminated environments or in cases of excess mortality of bees and decline in hives production. In addition, many epidemiological studies have suggested a correlation between the professional use of pesticides and the appearance of some pathologies in the concerned populations [2]. Carcinogenic effects, neurotoxic or endocrine disruption-type of pesticides have been highlighted in animals. The risk to humans is therefore issue both at the professional level or during consummation [3].
The brain is particularly vulnerable to oxidative damage because of its richness in peroxidable fatty acid, its high-energy demands, and its relatively low antioxidant power [1]. This irreversible molecular damage is the leading cause of neurodegenerative diseases [4].
In this context, the present study opted for the study of the pro-oxidant effects and neurobehavioral changes that can be induced following individually chronic exposure to an organochlorine incorporated ‘Acetamiprid’ which is a pesticide with the formula (C10H11ClN4) [5,6]. According to the toxicological information on the boxes of this last, they are hazardous and there is no specific antidote, that is why we applied a treatment of this toxicity by Quercetin as a pure polyphenol and synthetic organic substance [7].
Materials and Methods
Animals
The study was conducted on rats (Wister) albino males, from the Pasteur Institute of Algiers, weighing about 240 g to 260 g. Animals are divided into a control group and three treated lots. They have free access to water and food. The temperature of the pet store is maintained at 23°C, with a moderate humidity and a 12/12 photoperiod.
Choice of doses
Substances that will be used in this study are (Acetamiprid and Quercetin), the doses were selected by referring to results of recent work [5,7-10]; doses have been adjusted to meet the daily exposure rate. Rats were randomly divided into four groups of six (6) males each. The first group was served as the control who received only 0.5 ml of distillated water; the two other groups were treated PerOs with freshly prepared Quercetin solution with a single dose of 10 mg/Kg body weight for 90 days [9-11]. The third groups were treated with Acetamiprid at 3.14 mg/kg body weight per day for 90 days. The final groups were treated with Combination (Acetamiprid at 3.14 mg/kg/day and Quercetin 10 mg/Kg/day) for 90 days, each group received the treatment PerOs every day for 90 days between 9:00 am and 10:00 am.
Sacrifice of animals and brain removal
After 03 months of treatment, the animals were sacrificed by decapitation, the brain is removed and divided into five parts, one part used for the measurement of metabolites and stress parameters in the cytosol, and other parts kept in -80 for further studies.
Tissue preparation
Brain (1 g) was homogenized in 2 ml of buffer solution of phosphate buffered Saline 1:2 (w/v: 1g tissue with 2 ml PBS, pH 7.4). Homogenates were centrifuged at 10000xg for 15 min at 4°C and the resultant supernatant was used for the determination of glutathione, malondialdehyde, and glutathione peroxidase, glutathione-S-transferase and catalase activity.
Weight measurement
The weight measurement is performed on rats, day to day on a regular basis during the adaptation period and three months of treatment, using a sensitive balance (Aston®).
Determination of carbohydrates
The determination of total soluble carbohydrates was done in brain tissue of rats according to the Dubois method [12]. The calculation of the actual concentrations is done by the equation derived from the standard curve prepared from a stock solution of glucose.
Protein estimation
The total protein contents of various samples were determined according to the method of Bradford [13] using a bovine serum albumin as a standard.
Determination of lipids
Brain lipids are assayed according to the method of Goldsworthy [14], the spectrophotometric reading is performed at 530 nm wavelength. The calculation of the actual concentrations is performed from the calibration curve made from a stock solution prepared using sunflower oil.
Determination of reduced glutathione (GSH)
Brain glutathione level (GSH) was measured utilizing the method described by [15]. 0.2 ml of a sulfosalicylic acid (SSA) at 0.25%, centrifuged for 5 min A1000 t/min, add 1 ml of Tris HCl buffer + EDTA (0.02M), pH 9.6, mix and add 0.025 ml the 5,5`-dithiobis 2-nitrobenzoic acid (DTNB) 0.01M was dissolved in absolute methanol, measure the absorbance (A) at 412 nm.
Determination of glutathione-S-transferase (GST)
Glutathione-S-transferase (GST) catalyzes the conjugation reaction with glutathione in the first step of mercapturic acid synthesis. The activity of GST was measured according to the method of Habig [2]. The P-nitro benzyl chloride was used as substrate. The absorbance was measured at 340 nm at 30 s intervals for 3 min.
Determination of glutathione peroxidase (GPX)
Glutathione peroxidase (GPx) activity was measured by the procedure of Flohe and Gunzler [16]. The obtained supernatant after centrifuging of 5% liver homogenate at 1500×g during 10 min followed by 10000×g for 30 min at 4°C was used for GPx assay. 1 ml of reaction mixture was prepared which contained 0.3 ml of phosphate buffer (0.1 M, pH 7.4), 0.2 ml of GSH (2 mM), 0.1 ml of sodium azide (10 mM), 0.1 ml of H2O2 (1 mM) and 0.3 ml of liver supernatant. The reaction was terminated by addition of 0.5 ml 5% TCA after 15 min of incubation at 37°C. Tubes were centrifuged at 1500×g for 5 min and the supernatant was collected. 0.2 ml of phosphate buffer (0.1 M, pH7.4) and 0.7 ml of DTNB (0.4 mg/ml) were added to 0.1 ml of reaction supernatant. After mixing, absorbance was recorded at 420 nm.
Determination of catalase activity (CAT)
The catalase (CAT) activity was determined according to the method of Aebi [17]. The H2O2 decomposition rate was followed by monitoring absorption at 240 nm. One unit of CAT activity is defined as the amount of enzymes required to decompose 1 μmol of hydrogen peroxide in 1 min. the enzyme activity was expressed as μmol H2O2 consumed /min/mg protein.
Determination of malondialdehyde (MDA)
The lipid peroxidation level in brain homogenate was measured as malondialdehyde (MDA) which is the product of lipid peroxidation and reacts with Thiobarbituric acid (TBA) as a reactive substance (TBARS) to produce a red complex with an absorbance peak at 532 nm [18]. Thus, 125 μl of supernatant were homogenized by sonication with 50 μl of PBS, 125 μl of TCA–BHT (trichloroacetic acid-butylhydroxytoluene) in order to precipitate proteins, and then centrifuged (1000×g, 10 min, and 4°C). After wards, 200 μl of supernatant were mixed with 40 μl of HCl (0.6 M) and 160 ml of TBA dissolved in Tris, and then the mixture was heated at 80°C for 10 min. The absorbance of the resultant supernatant was obtained at 530 nm.
Data processing
The experiments data were analyzed using statistical software MINITAB (MINITAB 17.0 for windows 7, 8 and 10. MINITAB Ins. USA), and all statistical comparisons were made by means of t-test and values of P<0.05 were considered significant. Results are expressed as means ± SEM, and to better visualize the results the selected graph is the histogram using the Microsoft Excel 2016.
Results and Discussion
The question of the impact of neonicotinoids on health is necessary in view of the widespread contamination of the environment, water and impregnation of food. Although the state of science does not provide overall synthesis that these health impacts and studies continue on the subject, several publications are appearing risk of chronic effects of these products on human health.
Action of Quercetin, Acetamiprid and combination (Quercetin/Acetamiprid) on settings growths in rats for 90 days
The parameters of growth (total weight, weight gain, relative weight and tissue metabolites) in the brains of treated rats and control rats after 90 days of treatment are shown on the Tables 1 and 2 (Figure 1-4).
| Variable group | ||||
|---|---|---|---|---|
| Parameters | control | Quercetin | Acetamiprid | Combination |
| weight(g) | 246 ± 13.374 | 265 ± 17.098 | 224 ± 5.050*** | 254 ± 15.984 |
| weight Gain (g) | 42 ± 5.012 | 59 ± 7.016 | 21 ± 8.011*** | 53 ± 7.13 |
| Relative weight of brain (%) | 0.628 ± 0.012 | 0.63 ± 0.014 | 0.681 ± 0.019*** | 0.625 ± 0.013 |
| Results are expressed as mean ± SE (n=6) t-test was used for multiple comparisons. | ||||
Table 1: Body weight change (g) and relative weight (%) of the brain in control rats and rats treated with the Quercetin, the Acetamiprid and the combination (Q/Ac) after 90 days of treatment.
Action on cytosolic metabolites
| Variable group | ||||
|---|---|---|---|---|
| Parameters | Control | Quercetin | Acetamiprid | Combination |
| Proteins (mg) | 0.348 ± 0.0092 | 0.3489 ± 0.006 | 0.3502 ± 0.008 | 0.349 ± 0.008 |
| Carbohydrates (µg) | 36.627 ± 0.664 | 35.191 ± 0.606 | 55.927 ± 0.659*** | 36.265 ± 0.8214 |
| lipids (µg) | 448.936 ± 0.153 | 449.484 ± 0.169 | 444.911 ± 0.134** | 447.649 ± 0.096 |
| Results are expressed as mean ± SE (n=6) t-test was used for multiple comparisons. ****P<0.0001, ***P<0.001, **P<0.01, *P<0.05 statistical significant as compared to control | ||||
Table 2: Change of some biochemical parameters (Proteins, carbohydrates and fats) in the brain in control rats and rats treated with Quercetin, the Acetamiprid and combining Quercetin/Acetamiprid after 90 days of treatment.
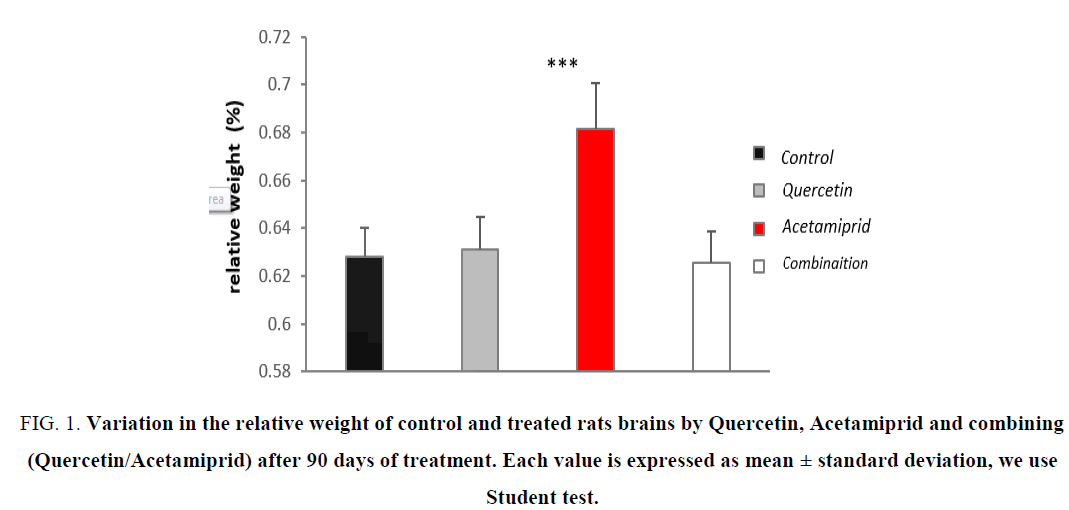
Action on relative weight of brain
Figure 1: Variation in the relative weight of control and treated rats brains by Quercetin, Acetamiprid and combining (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test.
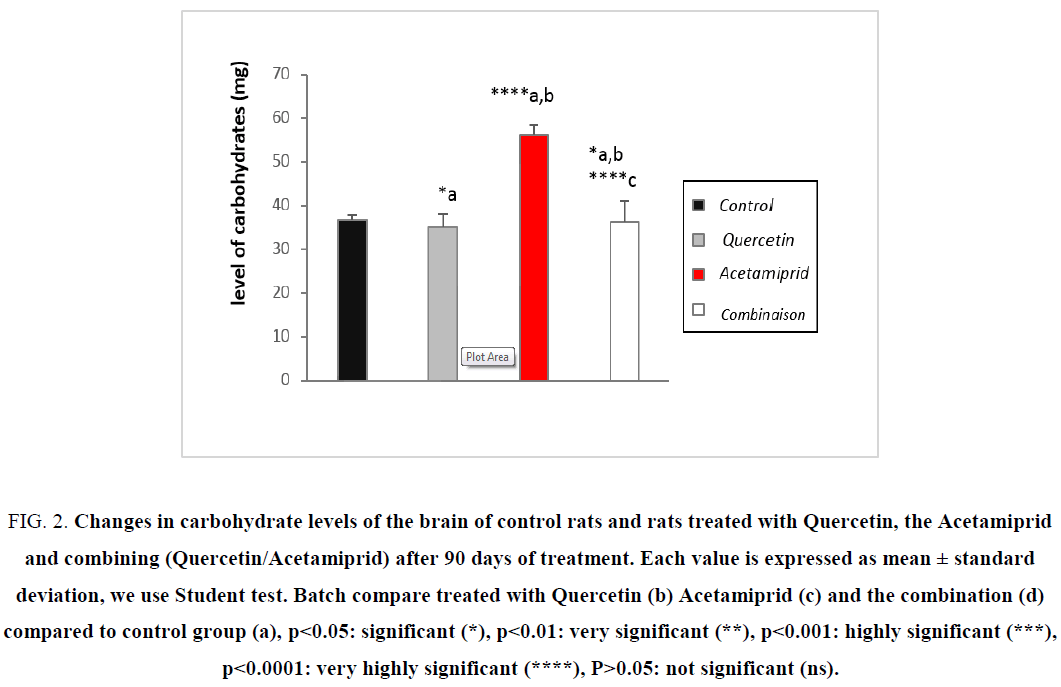
Action on the level of cytosolic carbohydrates
Figure 2: Changes in carbohydrate levels of the brain of control rats and rats treated with Quercetin, the Acetamiprid and combining (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a), ?<0.05: significant (*), ?<0.01: very significant (**), ?<0.001: highly significant (***), ?<0.0001: very highly significant (****), P>0.05: not significant (ns).
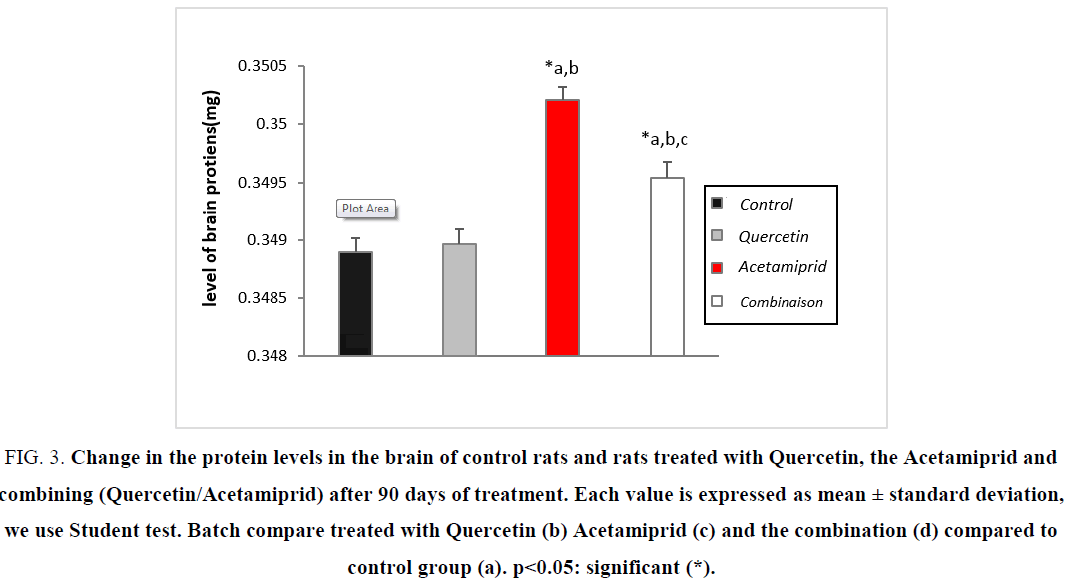
Action on the tissue proteins level
Figure 3: Change in the protein levels in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combining (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*).
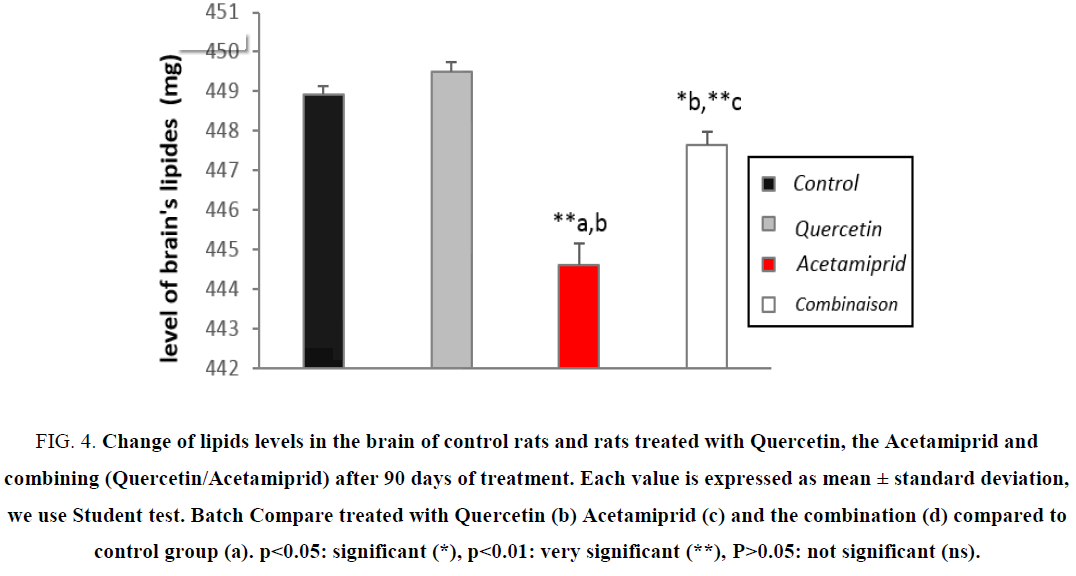
Action on the level of total lipids
Figure 4: Change of lipids levels in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combining (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch Compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*), ?<0.01: very significant (**), P>0.05: not significant (ns).
Our study is conducted by an experiment in albino rats to see the effect of a dose of (10 mg/kg/day) of Quercetin [8,9,19,20] on a dose of (3.14 mg/kg/j) Acetamiprid [5,6,9].
The results or experiment suggest that the administration of Acetamiprid as stressor by what an insecticide reduces body growth compared to controls (Figure 1-4).
Disturbance of metabolism, which corresponds to the stress response, is observed very frequently and translates the systemic effects of the release of pro-inflammatory cytokines and myoactives as modifications endocrine characteristics [1].
During the period of brain growth, the human brain depends on glucose as rat brain uses both glucose and ketone compounds to cover its energy and biosynthetic requirements [21].
Logically, Acetamiprid malnutrition caused by their psychic effects regarding the reduction of appetite [1], the latter produces a cellular stress disorder by the contribution in decreasing of physiological antioxidant system cofactors (vitamins, trace elements...) after the gavage dose of 3.14 mg/kg/day of Acetamiprid the total weight of the rats is reduced because low food intake this causes a malnutrition.
In contrast, the brain dominance used a strategy to keep and increase the half-life of nervous cells by several mechanisms unlike other guard cells, brain metabolism is overactive in case of stress, specifically, Acetamiprid has activated indirectly the β-oxidation in the brain, so the brain cytosolic lipid rate is decreased, the cell energy needs are recovering, this latest action confirmed by the rate of increase in cytosolic carbohydrates [4,21,22].
Because the nervous cells use fat as a primary source of energy on the one hand, and on the other hand, many stress disorder catalysis of enzymes and mechanism are enable to decrease or neutralize the damage [23-25], it is also translated into an increase of protein levels. The increase in relative weight (0.05%) is the result of total decrease in body weight and the increase of brain weight. after gavage of combination (Quercetin 10 mg/kg/day and Acetamiprid of 3.14 mg/kg/day), the antioxidant effect of the polyphenol is clearly present by the neutralization of the harmful effects of Acetamiprid due mainly by the increasing of appetite and setting cofactors and essential nutrients to the antioxidant system.
Our results are consistent with historical data [20,26,27] which showed that administration of Quercetin in rats treated with Acetamiprid which leads to the reduction of toxic effects.
All of these results shows that Quercetin appears to protect the brain against harmful or toxic effects of Acetamiprid. This weight gain gradually decreases from one week to the next throughout the period of taking Acetamiprid orally. The results clearly showed that Acetamiprid caused a marked decrease in food intake that is the cause of an anorectic effect. These results are consistent with the work of [26,27] who observed a reduction in food consumption in male rats in a sub-chronic accessed.
The results showed an impregnation of Acetamiprid in the brain, so the ability of crossing the blood brain barrier and act on the brain directly. These results justify the action of Acetamiprid on the cerebral antioxidant system in Wistar rats.
Action of Quercetin, of Acetamiprid and combination Quercetin/Acetamiprid on cytosolic parameters of oxidative stress in the brain after 90 days
Oxidative stress parameters (GSH, GPx, GST, CAT and MDA) in the brains of treated rats and control rats after 90 days of treatment are shown in the Table 3.
| Variable group | ||||
|---|---|---|---|---|
| Parameters | control | Quercetin | Acetamiprid | combination |
| GSH (nmol/mg proteins) | 140.55 ± 1.676 | 145.56 ± 1.366*a | 91.37 ± 1.160****a,b | 132.86 ± 1.58**a, b***c |
| GPx (µmol/mg proteins) | 0.243 ± 0.014 | 0.248 ± 0.017 | 0.188 ± 0.012**a***b | 0.229 ± 0.006*a**b***c |
| GST (nmol/min/ mg prot) | 0.872 ± 0.0038 | 0.854 ± 0.0016*a | 0.995 ± 0.0007****a,b | 0.883 ± 0.0006*a**b****c |
| CAT (nmol/min/ mg prot) | 17.453 ± 0.130 | 18.719 ± 0.086*a | 11.641 ± 0.089****a,b | 16.893 ± 0.071**a,b*** *c |
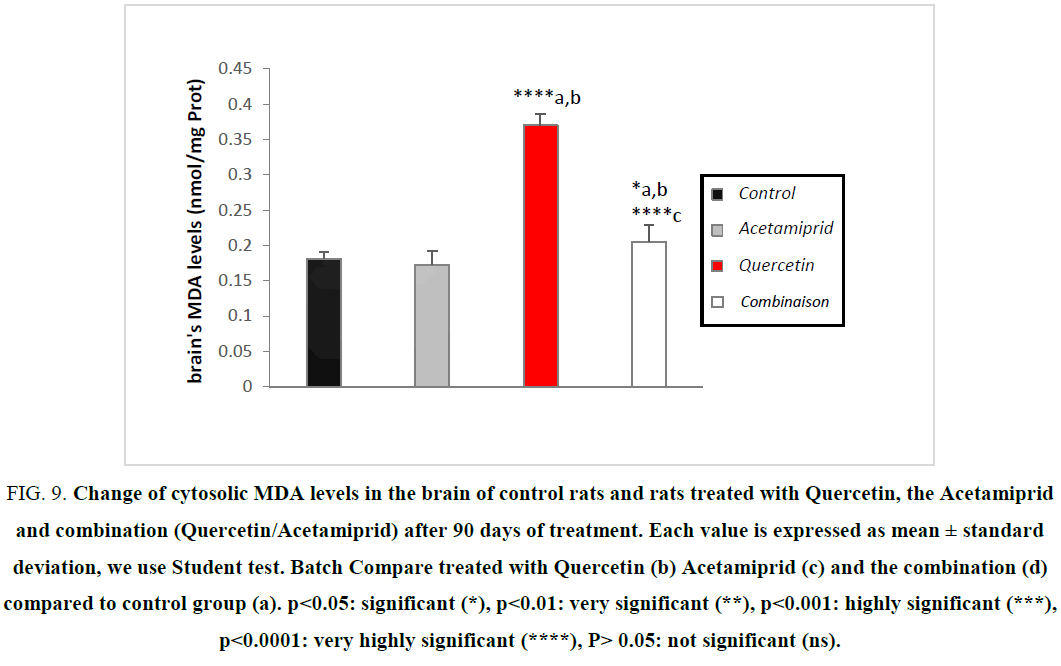
| MDA (nmol/mg prot) | 0.181 ± 0.010 | 0.173 ± 0.019 | 0.370 ± 0.016****a,b | 0.205 ± 0.023*a, b****c |
| Results are expressed as mean ± SE (n=6) t-test was used for multiple comparisons. ****P<0.0001, ***P<0.001, **P<0.01, *P<0.05 statistical significant as compared to control. | ||||
Table 3: Change in oxidative stress parameters GSH, MDA, GPx, GST and catalase in the brain in control rats and rats treated with the Quercetin, the Acetamiprid and the combination (Q/Ac) after 90 days of treatment.
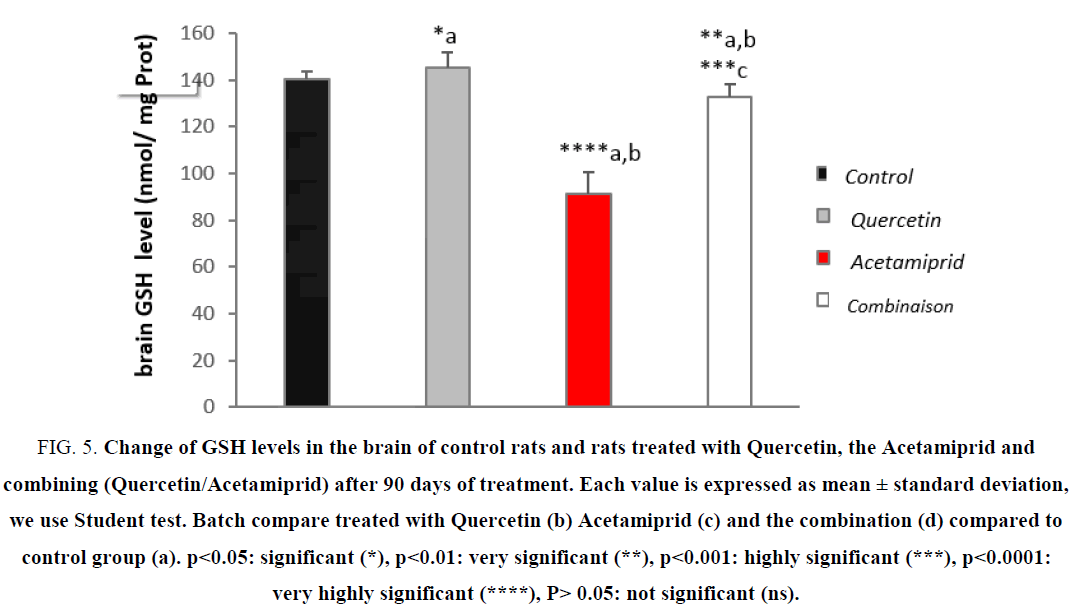
Evaluation of GSH level
GSH is a tripeptide play as the first defense against stress and thus considered as an essential compound that maintain cellular integrity due to its reducing property and its active participation in cellular metabolism [24,28].
Some of the important roles of glutathione are reduction or inactivation of ROS in the formation of glutathione disulfide (GSSG) and the conjugation of reduced glutathione (GSH) for the elimination of xenobiotics [29]. The results of our study are shown in Figure 5.
Figure 5: Change of GSH levels in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combining (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*), ?<0.01: very significant (**), ?<0.001: highly significant (***), ?<0.0001: very highly significant (****), P> 0.05: not significant (ns).
The study of oxidative stress parameters in tissues shows a very highly significant decrease of reduced glutathione (GSH) in the brain of the control group compared to the other groups.
Furthermore, the GSH level is decreased due to the high level of superoxide and free radicals GSH is converted to GSSG (oxidized glutathione) [15,30]. Treatment with Quercetin has restores significantly the GSH level compared to treated rats this is in accordance with the work of [20,26,27] on different pesticides. The Acetamiprid causes a decrease of GSH in the brain tissue leading to oxidative stress, while Quercetin increases this concentration [8,19]. These results are consistent with the work [2,20,31], they suggest that the reactive metabolites of a xenobiotic cause an increase in the levels of intracellular Ca+2.
That is accompanied with a decrease in GSH levels, it is mentioned that the alteration of calcium homeostasis generates mitochondrial oxidative stress which causes the formation of ROS, these last are depleting the cytosolic GSH [32,33]. results of a study on the kidneys treated with diesel, showed a significant increase of GSH, which is induced by the high levels of ROS generated by diesel constituents including PAHs [2,19,34], these results are confirmed the enzyme inhibition power Acetamiprid in the brain at doses of 0.34 mg/kg/d.
According to Kumar et al. found a decrease in glutathione levels in rats treated with B(a)P, which may be due to the extensive use of glutathione by enzymes, and a reduction in the activity of glutathione synthesis enzymes such as the enzyme glucose-6-phosphate, and glutathione peroxidase, and the neutralization of the hydroxyl radicals and singlet oxygen.
Although mitochondria produces continuous ROS during oxidative phosphorylation, leaking from superoxide in the electron transport chain is scanned by mitochondrial and cytoplasmic superoxide dismutase (SOD) and peroxidases GSH by reducing the rate of GSH [3,26,27]. The smoked cigarette continent many electrophiles and ROS also depletes GSH levels in the alveolar epithelial cytosolic cells in vitro and in vivo in the lung [24].
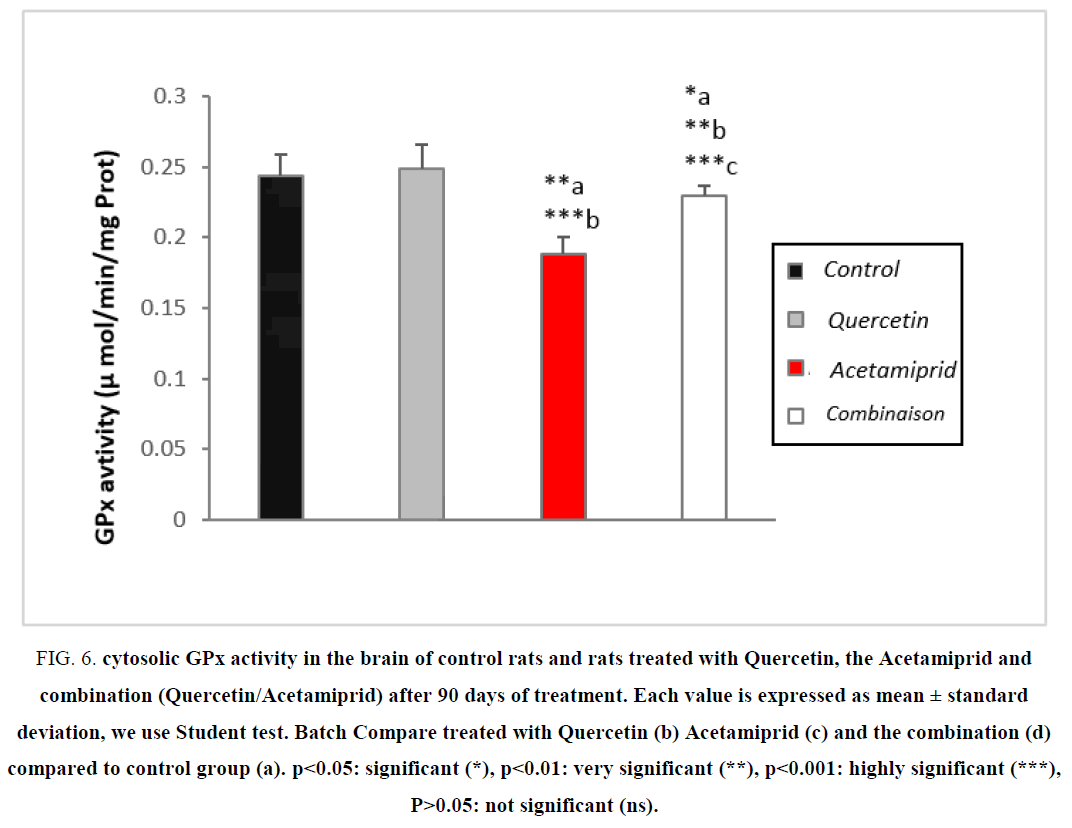
Evaluation of the enzymatic activity of GPx
The antioxidant enzymes are considered the first bodyline defense against free radicals, where they prevent the oxidation of biological macromolecules [16].
According to the Table 3 and Figure 6 the treatment of rats by Acetamiprid caused a highly significant (P>0.001) decrease of the enzymatic activity of glutathione peroxidase (GPx) activity in the brain compared to controls. However, there has been a highly significant increase of glutathione peroxidase (GPx) in rats treated with the combination compared to the treated with Acetamiprid [30,35].
Figure 6: Cytosolic GPx activity in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combination (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch Compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*), ?<0.01: very significant (**), ?<0.001: highly significant (***), P>0.05: not significant (ns).
GPx is an antioxidant key enzyme which regulates the level of ROS (GPx is capable of not only reducing the hydrogen peroxide in water, but also the oxidation of unsaturated fatty acids hydroperoxides) and thus protects the cells against damage generated by the toxic dose of Acetamiprid. Moreover, according to our results there is a decrease in brain GPx activity in rats treated with Acetamiprid. This decrease is mainly due to an overproduction of hydrogen peroxide and depletion of Quercetin and GSH in detoxification of Acetamiprid [27]. Resulting in a decrease of GPx activity [1].
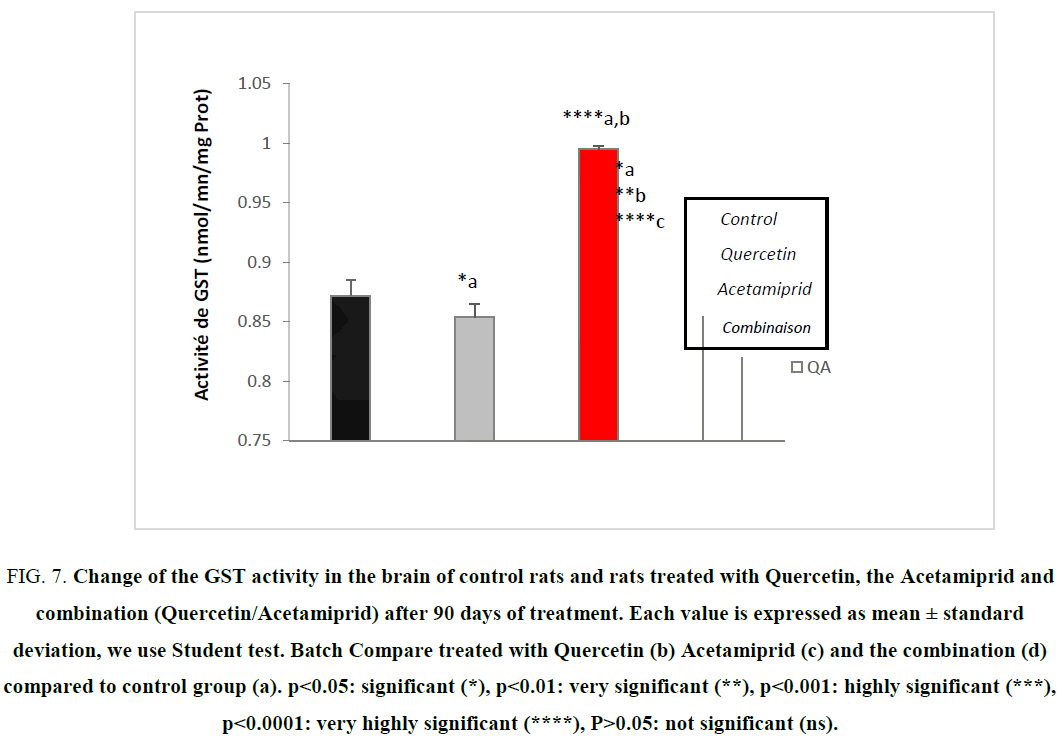
Evaluation of the enzymatic activity of GST
The GST are essentially cytosolic family of multifunctional enzymes, involved in various operations of transport and intracellular biosynthesis [24,30,35]. However, the most studied function of GST as regards the prevention of environmental xenobiotic, their dwelling activity of catalyzing conjugation reactions between a peptide, GSH and reactive molecules containing electrophilic sites capable of reacting dangerously with macromolecules such as nucleic acids. Catalyzing the conjugation of glutathione with certain substrates represents a step in the formation of compounds that are less toxic and more water soluble than the starting molecules which promotes their elimination from the body [24,29,36]. GST is also involved in the reduction of ROS damages in different cells.
Our study allowed us to measure the activity of this enzyme in the brain cells after a stress-induced by Acetamiprid. Figure 7 showed the variation of the enzymatic activity of cytosolic GST following an Acetamiprid administration.
Figure 7: Change of the GST activity in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combination (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch Compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*), ?<0.01: very significant (**), ?<0.001: highly significant (***), ?<0.0001: very highly significant (****), P>0.05: not significant (ns).
A very highly significant (p<0.000) of enzyme activity was observed in the group treated by Acetamiprid to (3.14 mg/kg/day).
The results of our study are consistent with previous studies that have shown that exposure to family of xenobiotics can induce GST activity [20,27]. The combination of their reactive metabolites with GSH in the GST may inactivate the compound and prevent the formation of adducts with DNA and mutagenicity. Therefore, the increase in the expression of GST is generally observed when the cells are under stress [30].
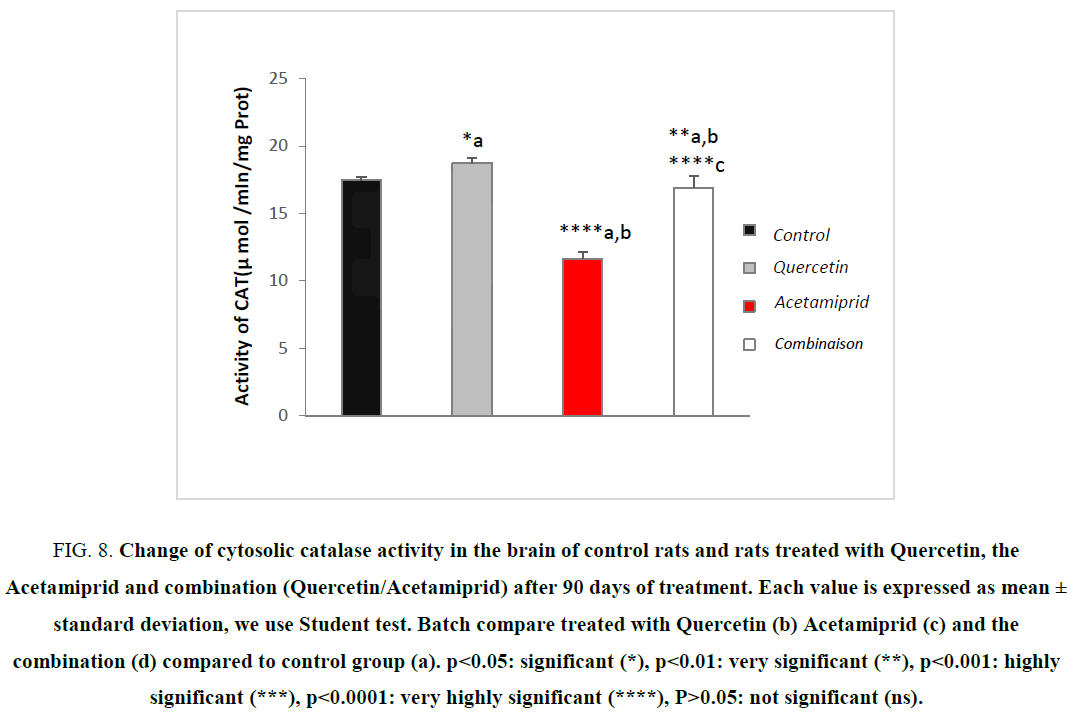
Evaluation of the enzymatic activity of catalase
The activity of catalase is specifically subservient to the conversion of the hydrogen peroxide into oxygen and water [37]. It is mainly located in the peroxisome. It thus prevents oxidative damages within the peroxisome, and prevents its propagation in the rest of the cell [18].
In this study, we intend to assess the opposite effect of Quercetin on the toxicity of Acetamiprid on enzymatic activity of catalase.
Figure 8 shows the variation of the enzymatic activity of cytosolic catalase between the control group, and other groups.
Figure 8: Change of cytosolic catalase activity in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combination (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*), ?<0.01: very significant (**), ?<0.001: highly significant (***), ?<0.0001: very highly significant (****), P>0.05: not significant (ns).
The results of this study showed a very highly significant decrease in cytosolic catalase of the brain in rats treated with Acetamiprid. This result suggests that Acetamiprid indirectly induces an increase in H2O2, so it is a case of causing oxidative stress. However, treatment with the (pesticide/Quercetin) presented an improvement on treatment with the pesticide only by the radical OH° is a possible candidate. Indeed, the decrease in CAT and GPx activity causes an increase in H2O2 that can produce OH° radical [38,39]. This is explained by a depletion of this enzyme following repeated exposure of Acetamiprid which generates of ROS during its metabolism including derivatives Quinones, an increase of SOD, a parameter that is not considered here, leads to an increase of H2O2 production, it also causes reduction of catalase [9,37]. These results disagree with several authors have tested the effect of some types of pesticides leading to the increase in catalase activity in animals, because of the existence of these pesticides in the body and subsequently boosting antioxidant defense [26,27,32].
All of these results showed that the dose of (10 mg/Kg/day) of Quercetin seems to protect the brain by increasing the antioxidants enzymatic activities in this experiment.
Evaluation of MDA levels
Because of the difficulty of measuring free radicals (reactive intense, extremely short half-life, low concentration); indirect markers are determined [23]. thiobarbituric acid is a powerful alkylating agent capable of reacting with biological macromolecules [40]. MDA levels is proportional to the extent of lipid peroxidation [41], it is considered as one of the fundamental mechanisms of cell damage caused by free radicals. Indeed, the lipid peroxidation resulting in the denaturation of polyunsaturated fatty acids component membranes. Thus, when a stress caused by the introduction of a contaminant such as pesticides, when the antioxidant systems become ineffective, cell damage can be estimated by the MDA levels. Therefore, it is a good biomarker for the measurement of oxidative stress.
During our work, we recorded a very highly significant increase in MDA levels in the brain of treated rats compared with controls Figure 9. This result is consistent with other studies [20,23,27,28,32,36].
Figure 9: Change of cytosolic MDA levels in the brain of control rats and rats treated with Quercetin, the Acetamiprid and combination (Quercetin/Acetamiprid) after 90 days of treatment. Each value is expressed as mean ± standard deviation, we use Student test. Batch Compare treated with Quercetin (b) Acetamiprid (c) and the combination (d) compared to control group (a). ?<0.05: significant (*), ?<0.01: very significant (**), ?<0.001: highly significant (***), ?<0.0001: very highly significant (****), P> 0.05: not significant (ns).
An increase in lipid peroxides indicate serious damage to the cell membrane, inhibition of several enzymes, cell function and cell death [1,41-43].
In the end after Quercetin feeding at a dose of 10 mg/kg/day in rats treated with Acetamiprid for a period of 90 days, a recovery state on the different stress parameters was observed, knowing that Quercetin is one of several active herbal extract play an important role in the prevention of various diseases related to oxidative stress [11,43] and regulates the activity of a variety of enzymes and cellular receptors, the work of [9,38] showed that the quercetin extract induced changes on the composition of lipids of cells, antioxidant status and enzymes attached to the membrane, which is a direct or indirect consequence of oxidative stress.
Conclusion
The study clearly demonstrated the subchronic exposure to Acetamiprid used in this study was able to induce neurotoxicity by abnormalities and a state of oxidative stress in total brain areas in adult male rats. Furthermore, the feeding of Quercetin to treated rats showed improvements affected all parameters.
Acknowledgement
Many thanks and acknowledgement are sent to Professor Said Fekra rector of Tebessa university for his help and facilities to work in the laboratories, also to all toxicology laboratory members of Tebessa university.
References
- Rouabhi R. Fungicides. Carisse O, editor. In Tech, Publishing. 15, ISBN: 978-953-307-266-1, 2010.
- Habig WH, Pabst MJ, Jakoby WB. Glutathione-S-transferase the first step in mercapturic acid formation. J Biol Chem. 1974;36(249):71309.
- Calderón-Segura ME. Evaluation of genotoxic and cytotoxic effects in human peripheral blood lymphocytes exposed in vitro to neonicotinoid insecticides news. J Toxicol. 2012;14:17.
- Adams J, Vorhees CV, Middaugh LD. Developmental neurotoxicity of anticonvulsants: human and animal evidence on phenytoin. Neurotoxicol Teratol. 1990;1(12):203-14.
- Scientific Opinion on the developmental neurotoxicity potential of acetamiprid and imidacloprid. EFSA J. 2013;11(12):3471.
- Ghersi-Egea JF, Livertoux MH, Minn A, et al. Enzyme mediated superoxide radical formation initiated by exogenous molecules in rat brain preparations. Toxicol Appl Pharmacol. 1991;32(110):107-17.
- Joint FAO/Who meeting on pesticide residues. Acceptable daily intakes, short-term and long-term dietary intakes, FAO/WHO, 2015;39:09-33.
- Gregory S, Kelly ND. Quercetin. Alt Med Rev. 2011;16(2):172-94.
- John C, Quindry, MCAnulty RS, et al. Oral quercetin supplementation and blood oxidative capacity in response to ultramarathon competition. Int J Sport Nutr Exer Met. 2008;38(18):601-16.
- Junko KK, Yukari K, Yoichiro K, et al. Nicotine-like effects of the neonicotinoid insecticides acetamiprid and imidacloprid on cerebellar neurons from neonatal rats. PLoS ONE. 2012;7(2):e32432.
- Nakamura Y, Ishimitsu S, Tonogai Y. Effects of quercetin and rutin on serum and hepatic lipid concentrations, fecal steroid excretion and serum antioxidant properties. J Health Sci. 2000;46(4):229-40.
- Dubois M, Gilles K, Hamiltona JK, et al. Colorimetric method for determination of sugars. Nature. 1956;168(4265):167.
- Bradford M. A rapid and sensitive method for the quantities of microgram quantities of protein utilizing the principle of protein-dye binding. Ann Biochem. 1976;11(72):248-54.
- Kaplan A. Lipids. Clin Chem. 1984;41:918-9.
- Weckbercker G, Cory JG. Ribonucleotide reductase activity and growth of Glutathione-depended mouse Leukemia L1210 cells in vitro. Can Letter. 1988;40(3):257-64.
- Flohe G. Analysis of glutathione peroxidase. Methods Enzymol. 1984;29(105):114-121.
- Aebi H. Catalase. Methods in enzymology. 1984;3(105):121-6.
- Fleming L, Mann JB, Bean J, et al. Parkinson’s disease and brain levels of organochlorinepesticides. Ann Neurol. 1999;4(36):100-3.
- Buening MK, Chang RL, Huang MT, et al. Activation and inhibition of benzo(a)pyrene and aflatoxin B1 metabolism in human liver microsomes by naturally occurring flavonoids. Cancer Res. 1981;13:67-72.
- Chen X, Ahn DU. Antioxidant activities of six natural phenolics against lipid oxidation induced by Fe2+ or ultraviolet light. JAOCS. 1998;75(12):1717-21.
- Nehlig A. Cerebral energy metabolism, glucose transport and blood flow: changes with maturation and adaptation to hypoglycaemia. Diabetes Met. 1997;44(23):18-29.
- Pellerin L. Activity-dependent regulation of energy metabolism by astrocytes: An update. Glia. 2007;46(55):1251-62.
- Adegbesan B, Adenuga G. Effect of lead exposure on liver lipid peroxidative and antioxidant defense systems of protein-undernourished rats. Biol Trace Elem Res. 2007;2(116):219-25.
- Sauer E. Liver delta-aminolevulinate dehydratase activity is inhibited by neonicotinoids and restored by antioxidants agents. Int J Environ Res Public Health. 2014;11(11):11676-90.
- Pomierny CL. Oxidative stress biomarkers in some rat brain structures and peripheral organs underwent cocaine. Neurotox Res. 2013;23:92-102.
- Benzidane C. Effets toxiques des pesticides, these magister. Univ sétif. 2012;8:73.
- Bourbia S. Evaluation of toxicity of pesticide mixtures on soil contamination bioindicator Helix aspersa, These PhD. Annaba University. 2013;10:177.
- Deneke SM, Fonburg BL. Regulation of cellular glutathione. Am J Physiol. 1988;257: Ll6373.
- Di-Monte DA, Lavasani M. Environmental factors in Parkinson’s disease. Neurotoxicology. 2002;23:487-502.
- Di-Monte DA, Chan P, Sandy MS. Glutathione in Parkinson’s disease: A link between oxidative stress and mitochondrial damage. Ann Neurol. 1992;22(S1):S111-5.
- Chen M1, Tao L, McLean J, et al. Quantitative analysis of neonicotinoid insecticide residues in foods: implication for dietary exposures. J Agric Food Chem. 2014;2:62(26):6082-90.
- Bear MF, Connors BW, Paradiso MA. Neuroscience: Exploring the Brain. 4th ed. New York; 2016;7(612.8-dc23):1019.
- Gibbons D, Morrissey C, Mineau P. Examination of the direct and indirect effects of neonicotinoids and fipronil on vertebrate fauna. About Sci Pollut Res. Springer. 2014;33.
- Icenogle LM. Behavioral alterations in adolescent and adult rats caused by a brief subtoxic exposure to chlorpyrifos during neurulation. Neurotoxicol Teratol. 2004;26:95-101.
- Bidlack WR. Interrelationships of food nutritiand health: The National Association of State Universities and Land Grant Colleges White Paper. J Am Coll Nutr. 1996;15:422-33.
- Esterbauer H, Gebicki J, Puhl H, et al. The role of lipid peroxidation and antioxidants in oxidative modification of LDL. Free Radic Biol Med. 1992;13:341-90.
- Federico A, Cardaioli E, da PozzoMitochondria P. Oxidative stress and neurodegeneration. Neurol Sci. 2012;322:254-62.
- Cory-Slechta DA, Thiruchelvam M, Richfield EK, et al. Developmental pesticide exposures and the Parkinson’s disease phenotype. Birth Defects Res Clin Mol Teratol. 2005;73:136-9.
- Banerjee BD, Seth V, Ahmed RS. Pesticide-induced oxidative stress: perspectives and trends. Rev Environ Health. 2001;16:1-40.
- Bashkatova V, Alam M, Vanin A, et al. Chronic administration of rote-none increases levels of nitric oxide and lipid peroxidation products in rat brain. Exp Neurol. 2004;186:235-241.
- Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1984;105:302-10.
- Rouabhi R, Djebar H, Djebar MR. Toxic effects of combined molecule from novaluron and diflubenzuron on paramecium. Am-Euras J Toxicol Sci. 2009;1(1):74-80.
- Rouabhi R, Djebar-Berrebbah H, Djebar MR. Impact of flufenoxuron, an IGR pesticide on Gallus domesticus embryonic development in ovo. J Cell Anim Biol. 2008;2(3):87-91.