Review
, Volume: 16( 3) DOI: 10.37532/0974-7435.2020.16(3).208Microfluidics- An Emerging Technology; Its Types and Applications
- *Correspondence:
- Isha Ishtiaq
Department of Biotechnology
University of Sialkot
Sialkot, Pakistan
Tel: 03247966429
E-Mail: ishaishtiaq@gmail.com
Received: August 03, 2020; Accepted: September 21, 2020; Published: September 28, 2020
Citation: Nayyab S, Ishtiaq I, Shahzadi S. Microfluidics- An Emerging Technology; Its Types and Applications. Biotechnol Ind J. 2020;16(3):208.
Abstract
Microfluidics has become the most sophisticated technology in the field of science and medicine. It exhibits several characteristics that attract the attention of researchers. The advantages of this fluid flow technology on miniaturized devices have urged scientists to study it. Microfluidic innovation is an empowering innovation for Lab-On-a-Chip (LOC) tests as it permits high portability and adaptability. Microfluidics has emerged as a powerful way to do it in vitro cell systems, sometimes called "organ-on-chip" or "body on a chip", which mimics the in vivo environment. There are many types of microfluidics being used in different areas of research. Applications of microfluidics are in the sector of medicine as well as in industry. Microfluidics has also been used in many biological assays, in which the most important and active area is nucleic acid amplification-based LAMP (Loop-Mediated Isothermal Amplification). This review describes some main types of microfluidics and its applications in various fields.
Keywords
Lab on a chip; Innovation; Amplification; LAMP; Biological assays
Introduction
In recent years, microfluidics has become one of the most interesting areas for researchers. It has garnered the attention of scientists due to its various advantages in clinical diagnostics as well as in other fields of science. It has practical applications in the design of systems that process low volumes of fluids to achieve multiplexing automation and high-throughput screening. Other microbiological assays that have been used are time-consuming and laborious [1]. Microfluidic devices are used mainly to improve analytical performance by performing miniaturization. Microfluidics has several advantages; it is a low-cost technique, it has precise control over low volume samples, it is rapid, it has a high surface to volume ratio and allows detection of various molecules on a small chip [2]. Microfluidic innovation is an empowering innovation for Lab-On-a-Chip (LOC) tests as it permits high portability and adaptability [2]. Microfluidics has emerged as a powerful way to do it in vitro cell systems, sometimes called "organ-on-chip" or "body on a chip", which mimics the in vivo environment [3]. Microfluidics has been used in many biological assays, such as electrophoresis, immunoassays, nucleic acid amplification analysis, cell manipulations, and so forth. Among these assays, nucleic acid amplification-based microfluidics is an active area of research. LAMP (Loop-Mediated Isothermal Amplification) is the most broadly used method for nucleic acid amplification [4]. LAMP is most sophisticated because it does not need regular thermal cycling, highly sensitive, convenient method and give rapid results.
Types of Microfluidics
There are many types of microfluidics i.e. digital microfluidics, paper microfluidics, cord-based microfluidics, diffusion-based microfluidics, plug-based microfluidics, inertial microfluidics, and droplet microfluidics. Microfluidics techniques also have been integrated with many other strategies.
Digital Microfluidics (DMF) RPA Assay
The Digital Microfluidics (DMF) is an alternative microfluidic molecular assay using liquid droplets and improves assay performance. It involved the method of Electrowetting on Devices (EWOD) to move liquids. In this report, DMF devices controlled by Thin-Film Transistor (TFT) electronics have been generated. Gene detection time and limit of detection increased on the AM-EWOD platform as compared to benchtop RPA assay. The capability of the DMF technique was exploited to amplify DNA from antibiotic-resistant bacteria in a triplex assay. The triplex assay involves the identification of 3 resistance genes i.e. NDM-1, KPC, and CTX-M-15 encoding carbapenems and ESBL respectively. Isothermal amplification method and fluorescence readout have been employed to identify these genes. DMF device was developed by using a TFT substrate and ITO (indium tin oxide) top plate along with an integrated temperature sensor [5].
Inertial Microfluidics
Using inertial microfluidics, microalgae named as Euglena have been separated based on shape. In inertial microfluidics, two inertial lift forces are applied and particles move to dynamic balance positions across liquid streamlines. The results showed high-throughput and label-free separation of microalgae cells [6]. Inertial microfluidics has also been employed for the separation of microalgae. Fluorescent polystyrene beads were used for the optimization of a microchannel. Microchannel was made up of 8 circular loops, 1 inlet, and 2 outlets. In this method, three major inertial forces were applied. The results proved this technique feasible and effective one [7].
Diffusion-Based Microfluidics
In bacteria, cell lysis was also analyzed by employing diffusion-based microfluidics. The lysis chip was fabricated of PDMS replica. This technique proved that the lysis solution and lysis time can be analyzed rapidly by changing flowrates. This research showed that microfluidic technology can be integrated with other microfluidics modules and effective in the future [8].
Tris Microfluidics
Interactions between the roots of plants and bacteria were also analyzed by using TRIS (Tracking Root Interactions System) microfluidics. TRIS device was fabricated by imaging and was integrated with microfluidics. As a result, real-time bacterial interactions were studied with high resolution. TRIS microfluidics can enhance the rate of discoveries in the field of science [9]. Contact dependent bacterial interactions were also determined onto PDMS microfluidic chip [10].
Drop Based Microfludics
Drop based microfluidic devices were used for the encapsulation of single cells. Cord based microfluidic cells, Fluorescence-Activated Cell Sorter FACS, and PDMS were used. We use microfluidic devices exemplify, hatch, and control singular cells in Picoliter watery drops in a transporter liquid at paces of up to a few hundred Hz [11].
Cord Based Microfluidics
Cord based microfluidic devices were used for glucose and ELISA assay. The chip materials utilized were Ucad (microfluidic cord based analytical devices), Horseradish Peroxidase (HRP), Potassium Iodide (KI). In this examination, biotinylated goat anti-mouse immunoglobulin (IgG) antibody, rabbit IgG antibody, and glucose are quantitatively detected [12].
Acuostofluidics
Acuostofluidics was employed to isolate exosomes from Extracellular Vesicles (EVs) using an integrated chip. In this method, acoustics and microfluidics techniques were combined and exosomes were isolated. Two modules were formed i.e. cell removal module and exosome-isolation module and acoustic radiation force were applied followed by stokes drag force. This label-free separation of exosome provided high purity and high yield [9].
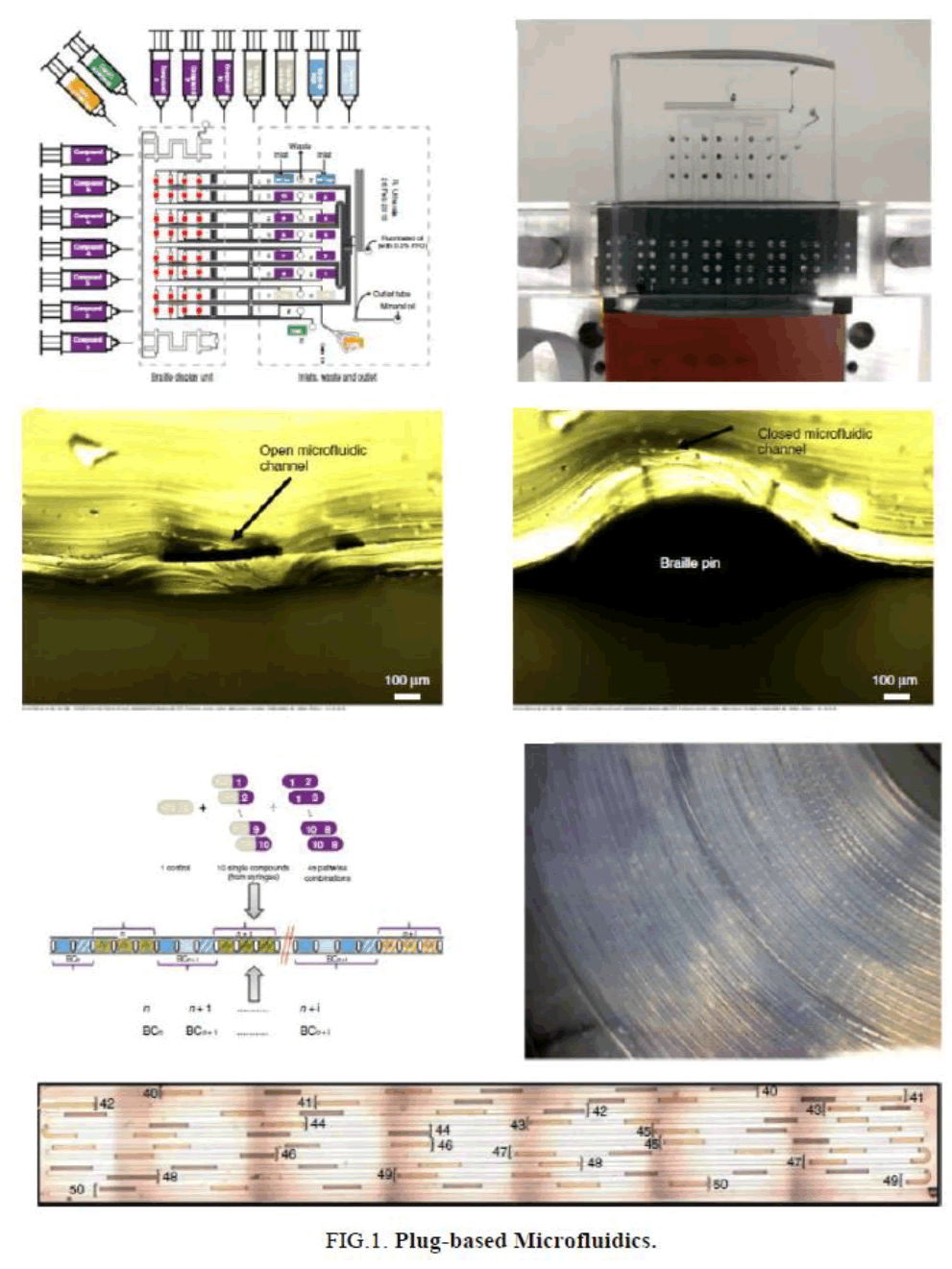
Plug-Based Microfluidics
Drug sensitivity has been determined by isolating CTCs with the ligand-receptor binding assay in a microfluidic device using a CTC chip [13]. Plug-based microfluidics has been used for combinatorial drug screening on cancer biopsies [14]. In this strategy, two-phase microfluidics was combined with braille valves and different types of plugs were used. Cancer cells have been isolated and quantified using microfluidics that provides the highest efficiency and purity [14,15].
Paper-Based Microfluidics (μpads)
Paper-based microfluidics (μPADs) is a non-invasive strategy to detect glucose and uric acid colorimetrically with great sensitivity. μPADS are disposable devices with reduced sample consumption and low-cost. μPADS were modified with polymer chitosan that is cheaper and faster to use [16]. μPAD has also been designed for dengue NS1 detection (DEN-NS1-PAD), which uses composite materials that are generally easy to find on the market. Paper-based devices such as cellulose paper were used to detect dengue NSI [17] (FIG. 1).
MITEV Device
TEVs were isolated using MITEV (Microfluidic Isolation of Tumor-Derived Extracellular Vesicles) device to detect mutation in the KRAS gene for diagnosis of Pancreatic Ductal Adenocarcinoma (PDAC) [18]. Microfluidic device (MITEV) can effectively absorb and release EVs from plasma in PDAC patients, in the short term (1.5 h collection and release data) in the TEV conversion study of the kidney. Each MITEV device is installed~100,000 equivalent zigzag columns and bundled in bulk layers before antibody synthesis, including two layers of nanoparticles that extend the surface of the device. Only one MITEV provided sufficient evDNA (total~2-14 ng from 2 ml plasma) to implement mutation detection using multiple platforms, such as droplet digital PCR or the nano String nCounter® platform. These two independent forums were used and it detected KRAS mutations within the isolated TEV treatment naïve PDAC patients. MITEV device may be used for maintenance applications, as the content of the discovery is at high-risk patients on examination, or routine examination dose capacity in patients with pancreatic cancer on treatment [18].
Padlock Probe Assay and C2CA Amplification
For the detection of mutations in codons, 12 and 13 of the KRAS gene Padlock probe assay and C2CA amplification were used. The chip material utilized as PMMA (Polymethylmethacrylate) and PDMA (Polydimethylsiloxane) [19].
Lamp (Loop-Mediated Isothermal Amplification)
Microfluidics also has been integrated with Loop-Mediated Isothermal Amplification (LAMP) assay which was used to detect Mycobacterium tuberculosis. The limit of detection was 1 pg/mL-1 and results showed high sensitivity and high specificity [20]. Prostate-Specific Gene (PSA) was also detected using LAMP on-chip. The efficiency of LAMP was 10-100 times higher than PCR [21]. LAMP has also been integrated with nanoprobe Au-Npsfor identification of the tlh gene for Vibrio parahaemolyticus. This strategy was easy, cheap, and rapid [22]. Salmonella keeps on being a significant health problem in Europe, just as in the remainder of the world. Our examination exhibited that microfluidics advancement is effective in the detection of food samples. One of the isothermal strategies that have great importance is Loop-Mediated Isothermal Amplification (LAMP). New methodologies have been created as of late with the point of improving unaided eye discovery of positive enhancement, for example, the utilization of gold nanoparticles. Results got from spiked nourishment demonstrated that hereditary focuses of SE performed well with exceptionally little contrasts. The chip material used was Polydimethysilioxane (PDMS) [1].
Combination of Microfluidic Loop-Mediated Isothermal Amplification with Gold Nanoparticles
A combination of Microfluidic Loop-Mediated Isothermal Amplification with Gold Nanoparticles is the best technique and it is used to detect Salmonella spp. in food samples. It was observed that Salmonella spp. was successfully detected by using this technique and successful DNA amplification was obtained. The chip material utilized was Computer-Numerical-Control (CNC) polymer machining, Polydimethylsiloxane (PDMS) replica molding, and PMMA for the detection of Salmonella spp. in food samples [2].
Passive Capillary Driven Microfluidic Chips
Flow rates assume a significant job in microfluidic devices because they affect the transport of chemicals and determine where and when (bio)chemical reactions occur in these devices. They decide the transaction between convection and diffusion, how much sample and reagents pass and may be consumed in a device, shear stress on objects for example cells in the microchannel, and influence blending and disintegration examples of reagents [23].
Applications of Microfluidics
Pumping Method is Necessary for Making Microfluidic Devices
An effective and easy to implement the pumping method is necessary for making microfluidic devices a ubiquitous commodity. The most important advantage of this pumping method is that it can be implemented in a wide variety of settings. For example, it can without much of a stretch be interfaced with high-throughput automated frameworks or utilized in research laboratories where much simpler equipment is available. The main hardware required is an equipment which can convey little amounts of fluid, for example, a syringe or pipette [24].
Microfluidics in Virology
Microfluidics technology has also been employed in the detection of viruses. Multiplex microfluidic loop-mediated isothermal amplification (mμLAMP) had been performed to detect multiple strains of influenza viruses. Microfluidics was integrated with LAMP and substrains of influenza and swine viruses were identified onto the chip efficiently with great specificity and high speed. The chip material used was Polydimethysilioxane (PDMS) with a good depth of 0.2 nm [25]. Infectious viruses had also been targeted by integrating bead-based RCA assay with microfluidics. It was sensitive and selective detection with reduced time of detection [26]. Microfluidic chip had been fabricated and integrated with a micro-magnetic field for detection and subtyping of multiple influenza viruses (H1N1, H3N2, H9N2). It was proved microfluidics is beneficial in terms of reduced sample consumption. It is a powerful technique for the detection of multiple viruses thus helpful in diagnostics [27]. Viral pathogens were isolated and characterized based on size by using a silicon nanowire forest combined with microfluidics. The results showed a high capture efficiency of pathogens because of the accurate inter-wire spacing. This was an effective method to detect and characterize viral pathogens [28,29]. The influenza H1N1 virus had been detected by performing a sandwich-based aptamers assay with integrated microfluidics. The results proved this technique efficient and effective for the detection of viruses. There was a decreased limit of detection and reaction time [30]. Pseudorabies Virus (PRV) also has been detected by using micro LAMP (μ-LAMP). The PDMS-Glass hybrid microfluidic chip was fabricated and results showed that μ-LAMP was highly specific. This method was more sensitive, specific, and less time consuming than other conventional methods [4]. Dengue Virus (DENV) infection is a significant health issue worldwide. We exhibit a μPAD designed for dengue NS1 detection (DEN-NS1-PAD), which uses composite materials that are generally easy to find on the market. Paper-based devices such as cellulose paper were used to detect dengue NSI. Dengue NS1 spiked in human serum was used for the optimization and characterization of the device [17].
Human Gut on A Chip Microdevice
A human gut-on-a-chip micro-device was utilized to co-culture multiple commensal microbes in contact with living human intestinal epithelial cells for more than seven days in vitro and to check how gut microbiome and peristalsis-associated mechanical deformations independently contribute to intestinal bacterial overgrowth and inflammation. The chip material utilized was PDMS polydimethylsiloxane. This human gut-on-a-chip can be utilized to break down the commitments of the microbiome to intestinal pathophysiology [31].
Isolation of Circulating Tumor Cells (CTCs)
One of the great features of microfluidics is its use in the isolation of Circulating Tumor Cells (CTCs) from peripheral blood. There are numerous methodologies used for this purpose i.e. PCR, affinity-based capture, ELISA but they have certain limitations. EpCAM-based strategies have been widely used for isolation of CTCs but the EpCAM receptor is not present on all cancer cells therefore this is not suitable for the detection of various cancers i.e. hepatocellular carcinoma [13]. One-step isolation of CTCs was performed by using the μ-MixMACS chip. The results showed that the μ-MixMACS chip is a powerful tool to isolate CTCs from various tumors without any change [32]. Microfluidics was employed to isolate CTCs with immunoaffinity based detection. In this method, DNA-assisted microfluidic chip was used that results in the efficient capture of CTCs [33]. Size-based isolation of CTCs was done by using a PMMA chip. Three cell lines; A549, SK-MES-1, and H446 were used as model and their capture rate was determined which was 95%. This was a rapid and cheap method thus helpful in cancer diagnosis and treatment [34]. CTCs have also been isolated using microfluidic chip integrated with wavy-herringbone micro-patterned surfaces. The capture efficiency of the wavy-HB chip was high than that of the grooved-HB chip [35].
Microfluidics in Food Industry
Food allergy is a major threat to health. Microfluidics has been used to detect allergens in food. Colorimetric LAMP microfluidic assay was performed to detect three types of allergens in food. It was a rapid, sensitive, and most convenient method [36]. Microfluidics ELISA-Based Optical sensors have also been developed for highly sensitive detection of food allergens [37]. A microfluidic system integrated with a Quantum dots (Qdots) aptamer functionalized Graphene Oxide (GO) nano-biosensor was also developed to detect peanut allergens in peanut or peanut products [38].
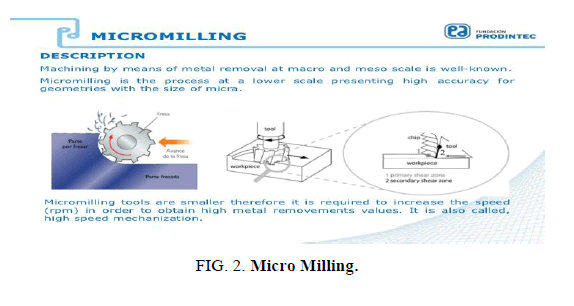
Micro Milling
A straightforward technique was developed to create bio-functional microfluidic channels using CO2 plasma to induce both carboxylic and hydroxyl groups onto the channel surface. We exhibit reconciliation of this procedure with micro-contact printing to covalently micro-pattern practical biomolecules inside microfluidic channels. The chip material utilized as Polydimethylsiloxane (PDMS). Recent work has shown that micro-milling is more effective for microfluidic devices. Micro-milling is a method that is used for ultra-rapid prototyping of plastic microfluidic devices. The chip material was used PDMS+Kit-On-A-Lid-Assays (KOALA) [39] (FIG. 2).
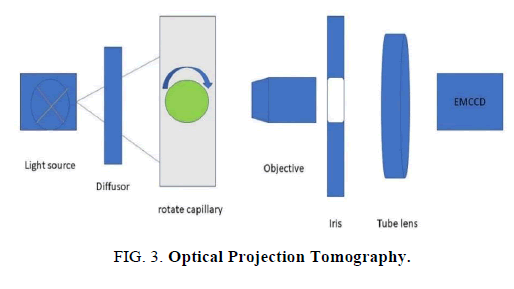
Optical Projection Tomography
OPT technology empowers three‐dimensional imaging of samples that are 1-10 mm in size. Samples of this size are enormous for confocal imaging, and little for Magnetic Resonance Imaging (MRI), but most vertebrate embryos are in this size range [40]. We design an OPT adaptor to perform OPT on a commercial wide‐field inverted microscope. Images from different angles were observed and analyzed [40] (FIG. 3).
Conclusion
Microfluidics have become the most refined innovation in the field of science and medication. Microfluidic development is an enabling advancement for Lab-On-a-Chip (LOC) tests as it allows high compactness and versatility. This paper depicts some principle sorts of microfluidics and its applications in different fields. Our research indicated that Microfluidics innovation is viable in detection of infections. Multiplex microfluidic circle interceded isothermal intensification (mμ LAMP) had been performed to identify numerous strains of influenza infections. In our study we use gene Padlock probe assay and C2CA amplification for the detection of mutations in codons 12 and 13 of KRAS. We discussed Microfluidics ELISA-Based Optical Sensor for high sensitive detection of food allergens. Recent studies have shown that micro-milling is more effective for microfluidic devices. It was concluded that microfluidics is a vast field that is very helpful in diagnostics and many other fields.
References
- Azinheiro S, Carvalho J, Prado M, et al. Evaluation of different genetic targets for salmonella enterica serovar enteriditis and typhimurium, using loop-mediated isothermal amplification for detection in food samples. Front Sust Food Syst. 2018;2:5.
- Garrido-Maestu A, Azinheiro S, Carvalho J, et al. Combination of microfluidic loop-mediated isothermal amplification with gold nanoparticles for rapid detection of Salmonella spp. in food samples. Front Microbiol. 2017;8:2159.
- Lu S, Dugan CE, Kennedy RT. Microfluidic chip with integrated electrophoretic immunoassay for investigating cell-cell interactions. Anal Chem. 2018;90(8):5171-78.
- Fang X, Liu Y, Kong J, et al. Loop-mediated isothermal amplification integrated on microfluidic chips for point-of-care quantitative detection of pathogens. Anal Chem. 2010;82(7):3002-6.
- Kalsi S, Sellars SL, Turner C, et al. A programmable digital microfluidic assay for the simultaneous detection of multiple anti-microbial resistance genes. Micromachines. 2017;8(4):111.
- Li M, Muñoz HE, Goda K, et al. Shape-based separation of microalga Euglena gracilis using inertial microfluidics. Sci Rep. 2017;7(1):1-8.
- Syed MS, Rafeie M, Vandamme D, et al. Selective separation of microalgae cells using inertial microfluidics. Bioresour Technol. 2018;252:91-9.
- Fradique R, Azevedo AM, Chu V, et al. Microfluidic platform for rapid screening of bacterial cell lysis. J Chromatogr A. 2020;1610:460539.
- Massalha H, Korenblum E, Malitsky S, et al. Live imaging of root-bacteria interactions in a microfluidics setup. PNAS. 2017;114(17):4549-54.
- Cooper R, Tsimring L, Hasty J. (2018). Microfluidics-based analysis of contact-dependent bacterial interactions. Bio- protocol. 2017;8(16).
- Köster S, Angile FE, Duan H, et al. Drop-based microfluidic devices for encapsulation of single cells. LOC. 2008;8(7):1110-5.
- Elomaa J, Gallegos L, Gomez FA. Cord-based microfluidic chips as a platform for elisa and glucose assays. Micromachines. 2019;10(9):614.
- Zhang Y, Zhang X, Zhang J, et al. Microfluidic chip for isolation of viable circulating tumor cells of hepatocellular carcinoma for their culture and drug sensitivity assay. Cancer Biol Ther. 2016;17(11):1177-87.
- Eduati F, Utharala R, Madhavan D, et al. A microfluidics platform for combinatorial drug screening on cancer biopsies. Nat Commun. 2018;9(1):1-13.
- Kokkinis G, Cardoso S, Keplinger F, et al. Microfluidic platform with integrated GMR sensors for quantification of cancer cells. Sens Actuators B: Chem. 2017;241:438-45.
- Gabriel EF, Garcia PT, Cardoso TM, et al. Highly sensitive colorimetric detection of glucose and uric acid in biological fluids using chitosan-modified paper microfluidic devices. Analyst. 2016;141(15):4749-56.
- Prabowo MH, Chatchen S, Rijiravanich P, et al. Dengue NS1 detection in pediatric serum using microfluidic paper-based analytical devices. ABC. 2020;1-11.
- Kamyabi N, Abbasgholizadeh R, Maitra A, et al. Isolation and mutational assessment of pancreatic cancer extracellular vesicles using a microfluidic platform. Biomed Microdevices. 2020;22(2):1-11.
- Ahlford A, Cond A, Sabourin D, et al. A microfluidic platform for personalized cancer diagnostics by padlock probe ligation and circle-to-circle amplification. In Proc. µTAS Conf. 2011;61-3.
- Rafati A, Gill P. Microfluidic method for rapid turbidimetric detection of the DNA of Mycobacterium tuberculosis using loop-mediated isothermal amplification in capillary tubes. Microchim Acta. 2014;182(3-4):523-30.
- Hataoka Y, Zhang L, Mori Y, et al. Analysis of specific gene by integration of isothermal amplification and electrophoresis on poly (methyl methacrylate) microchips. Anal Chem. 2004;76(13):3689-93.
- Kong C, Wang Y, Fodjo EK, et al. Loop-mediated isothermal amplification for visual detection of Vibrio parahaemolyticus using gold nanoparticles. Microchim Acta. 2018;185(1):35.
- Salva ML, Temiz Y, Rocca M, et al. Programmable hydraulic resistor for microfluidic chips using electrogate arrays. Sci Rep. 2019;9(1):1-10.
- Walker GM, Beebe DJ. A passive pumping method for microfluidic devices. LOC. 2002;2(3):131-4.
- Fang X, Chen H, Yu S, et al. Predicting viruses accurately by a multiplex microfluidic loop-mediated isothermal amplification chip. Anal Chem. 2011;83(3):690-95.
- Na W, Nam D, Lee H, et al. Rapid molecular diagnosis of infectious viruses in microfluidics using DNA hydrogel formation. Biosen Bioelectron. 2018;108:9-13.
- Zhang RQ, Hong SL, Wen CY, et al. Rapid detection and subtyping of multiple influenza viruses on a microfluidic chip integrated with controllable micro-magnetic field. Biosen Bioelectron. 2018;100:348-54.
- Xia Y, Tang Y, Yu X, et al. Label‐free virus capture and release by a microfluidic device integrated with porous silicon nanowire forest. Small. 2017;13(6):1603135.
- Xia Y, Zheng SY. A mcirofluidic device of biodegradable porous silicon nanowires for size based capturing and releasing viruses. In 2015 Transducers-2015 18th International Conference on Solid-State Sensors. Actuators and Microsystems (TRANSDUCERS) (pp. 444-447). IEEE. 2015
- Tseng YT, Wang CH, Chang CP, et al. Integrated microfluidic system for rapid detection of influenza H1N1 virus using a sandwich-based aptamer assay. Biosen Bioelectron. 2016;82:105-111.
- Kim HJ, Li H, Collins JJ, et al. Contributions of microbiome and mechanical deformation to intestinal bacterial overgrowth and inflammation in a human gut-on-a-chip. PNAS. 2016;113(1):E7-15.
- Lee TY, Hyun KA, Kim SI, et al. An integrated microfluidic chip for one-step isolation of circulating tumor cells. Sens Actuators B: Chem. 2017;238:1144-50.
- Chen D, Wen J, Zeng S, et al. DNA fragment‐assisted microfluidic chip for capture and release of circulating tumor cells. Electrophoresis. 2019;40(21):2845-52.
- Fan X, Jia C, Yang J, et al. A microfluidic chip integrated with a high-density PDMS-based microfiltration membrane for rapid isolation and detection of circulating tumor cells. Biosen Bioelectron. 2015;71:380-6.
- Wang S, Thomas A, Lee E, et al. Highly efficient and selective isolation of rare tumor cells using a microfluidic chip with wavy-herringbone micro-patterned surfaces. Analyst. 2016;141(7):2228-37.
- Sayad AA, Ibrahim F, Uddin SM, et al. A microfluidic lab-on-a-disc integrated loop mediated isothermal amplification for foodborne pathogen detection. Sens Actuators B: Chem. 2016;227:600-9.
- Weng X, Gaur G, Neethirajan S. Rapid detection of food allergens by microfluidics ELISA-based optical sensor. Biosensors. 2016;6(2):24.
- Weng X, Neethirajan SA. Microfluidic biosensor using graphene oxide and aptamer-functionalized quantum dots for peanut allergen detection. Biosen Bioelectron. 2016;85:649-56.
- Guckenberger DJ, de Groot TE, Wan AM, et al. Micromilling: a method for ultra-rapid prototyping of plastic microfluidic devices. LOC. 2015;15(11):2364-78.
- Du W, Fei C, Liu J, et al. Optical projection tomography using a commercial microfluidic system. Micromachines. 2020;11(3):293.