Original Article
, Volume: 17( 1)Pectin Transitions during Growth and Development of Bael (Aegle Marmelos (L.) Correa) Fruit
- *Correspondence:
- Anup KB, ICAR-Central Institute for Subtropical Horticulture, Lucknow-226 101, U.P., India, Tel: 48858318585; E-mail: dadabhatu@gmail.com
Received: May 04, 2017; Accepted: June 08, 2017; Published: June 12, 2017
Citation: Anup KB, Dikshit A, Dileep KT. Pectin Transitions during Growth and Development of Bael (Aegle Marmelos (L.) Correa)Fruit. Anal Chem Ind J. 2017;17(1):119.
Abstract
Quantitative and qualitative changes in the pectin fractions of two bael selections (CISH B-1 and CISH B-2) were estimated during fruit development between 150 and 345 days after fruit set (DAFS). The fruits were evaluated for alcohol insoluble solids (AIS), three pectin fractions (water, ammonium oxalate and alkali soluble), total soluble solids (TSS) and titratable acidity. An increase in TSS and titratable acidity in both the bael selections was noticed with the advancement of fruit development. Initially, Alcohol insoluble solids decreased during 150 to 210 DAFS, followed by a significant increase up to 335 DAFS, which decreased finally in fruits of CISH B-1 and CISH B-2 harvested at 345 DAFS. The total pectin did not change significantly throughout the growth period. The water-soluble pectin decreased initially in both the selections and increased significantly thereafter. However, alkali-soluble pectin showed an increasing trend initially and then decreased with the advancement of maturity in both the selections. The ammonium oxalate-soluble fraction of pectin remained low throughout the growth period in both the selections. The transition of pectin fractions (increase in water-soluble pectin and decrease in alkali-soluble pectin) in these two bael selections indicated that harvest maturity was attained around 300 DAFS.
Keywords
Bael; Aegle marmelos; AIS; Maturity; Pectin fractions
Introduction
Bael (Aegle marmelos (L.) Correa, family Rutaceae is a medicinally important indigenous fruit tree of Indian subcontinent. It grows in dry forests on hills and plains all over India (central India is the major growing area), southern Nepal, Myanmar, Pakistan, Bangladesh, Vietnam, Laos, Cambodia, Thailand, Malaysia, Sri Lanka, the Philippines and Fiji. All parts of the tree (leaves, fruits, stem, bark and roots) at all stages of development are found effective against many human ailments. The immature and unripe fruits are generally used as digestive, astringent, stomachic in the form of powder, murabba (preserve) and candy and are usually prescribed for diarrhoea and dysentery. In Indian traditional medicinal industry, ripe fruits are regarded as a tonic, a restorative, a laxative and good for the heart and brain and are processed into fruit bar, RTS, squash, etc. [1]. Bael fruit is one of the richest sources of many bioactive nutrients like marmelosin, psoralen, luvangetin, aurapten, marmelide, tannins, pectin, riboflavin, etc. [2].
Pectin is a nutritionally important but complex polysaccharide in plant cell walls with applications in food, pharmaceuticals and a number of other industries. Pectin has functions in plant growth, morphology, development and plant defense. It also serves as a gelling and stabilizing polymer in diverse food and specialty products, has positive effects on human health and multiple biomedical uses [3]. It is used to reduce blood cholesterol levels and gastrointestinal disorders. In the food industry, pectin is used in jams, jellies, frozen foods, and more recently in low-calorie foods as a fat and/or sugar replacer [4]. Isolation and characterization of pectic substances during development, maturity and ripening of various fruits like mango [5-7], guava [8,9], citrus [10], sweet cherry [11], cape-gooseberry [12], blueberry [13], Japanese quince [14], etc., have been reported in the literature [15-17]. Roy and Singh [18,19] reported the changes in total pectin and alcohol insoluble solids during development and ripening of bael fruit, but did not differentiate between pectin fractions. Two promising seedling selections of bael, CISH B-1 and CISH B-2, have been identified by the Institute (ICAR-CISH, Lucknow) through germplasm collection in which various nutraceutical parameters (including pectin and its fractions) need to be characterized before their release as varieties. Therefore, an attempt has been made to characterize pectin during the growth and development of these two selections to identify the proper harvesting period for processing purpose.
Materials and Methods
Plant materials
ICAR-Central Institute for Subtropical Horticulture, Lucknow has identified two promising seedling selections of bael, CISH B-1 and CISH B-2, through germplasm collection from the states of Uttar Pradesh and Bihar in India with high yield (42.6 kg/tree in B-1 and 38.5 kg/tree in B-2). The fruit of CISH B-1 is small in size (fruit weight varies between 0.8 to 1.2 kg) and oval to oblong in shape with good taste, less seed and thin rind. On the other hand, the fruit of CISH B-2 is larger in size (fruit weight ranges from 1.8 kg to 2.7 kg) and round to oblong in shape with thin rind, less fiber and few seeds [16]. Healthy fruit of both the selections was collected from the Institute farm at regular intervals (20 days) starting from 150 days after fruit set (second week of October) up to harvest (345 days after fruit set, first week of May). Six fruit from three trees (two fruit from each tree as one replication – total three replications) for each selection was harvested at each sampling date for this study. All the trees were 14 years old.
Experimental Procedure
The weight of individual fruit was recorded using an open pan balance with maximum capacity of 5 kg. Total soluble solids (TSS) of juice, obtained by squeezing fruit mesocarp through a fine muslin cloth, were measured by using hand refractometer (Erma, Japan). While titratable acidity of water extract of mesocarp was estimated by titrimetric method using 0.1 N sodium hydroxide (NaOH) solution with citric acid as reference standard [17]. The fruits were also evaluated for alcohol insoluble solids (AIS). AIS was then characterized into three pectin fractions using distilled water, 0.05 M ammonium oxalate and 0.05 M NaOH by subsequent extraction as per the method described in literature [5]. The total pectin was arrived by adding all the three fractions. AIS were estimated by taking 30 g bael mesocarp (without seeds) in a 250 ml conical flask and adding 100 ml of 95% alcohol to it. The mixture was boiled for 15 min, cooled to room temperature and filtered through Buchner funnel using Whatman no. 1 filter paper. The residue was washed with alcohol followed by acetone until it became white/colorless and then dried at 45°C till constant weight. Water-soluble pectin (WSP) was extracted from AIS by dispersing 0.5 g of AIS in 40 ml of distilled water, keeping in a water bath at 30°C for 2 h with intermittent shaking and finally centrifuging at 10,000 rpm (8944 xg) for 10 min. The residue was extracted again by repeating the same process.
The supernatants were combined and volume was made up to 100 ml in a volumetric flask. The left-over residue of AIS after WSP extraction was extracted with 0.05 M ammonium oxalate following the above procedure twice and making the final volume up to 100 ml to get ammonium oxalate-soluble pectin (OSP). Similarly, the residue left after ammonium oxalate extraction was further extracted with 0.05 M NaOH in the same way as above and volume was made up to 100 ml to obtain alkali-soluble pectin (ASP). All the pectin fractions were estimated colorimetrically by adding 1.0 ml of carbazole reagent (0.1% in absolute alcohol) and 12.0 ml of concentrated sulfuric acid to 2.0 ml of pectin solution with constant agitation. The tubes were closed with rubber stoppers, allowed to stand for 10 min and color was read at 525 nm in a double beam UV-VIS spectrophotometer (Labomed Inc., USA). Blank sample was prepared by adding 1 ml of purified alcohol instead of 1 ml carbazole in the reaction mixture. Standard solution was made using galacturonic acid (100 μg/ml).
Statistical analysis
The statistical analysis (CD at P<0.05) was carried out using the statistical software developed at Chaudhary Charan Singh Haryana Agricultural University, Hisar, Haryana, India [18]. Single-factor analysis using completely randomized design is followed in the calculation with two fruits as one replication (three replications for each selection for each harvest date) and student’s t-test was used for comparison of means. Two measurements per fruit sample were taken into consideration for statistical analysis.
Results and Discussion
Changes in fruit weight, TSS and titratable acidity
In CISH B-1, the average fruit weight increased from 954 g at 150 days after fruit set (DAFS) to 1040 g after 345 days of fruit set, while in CISH B-2 it increased from 1366 to 2573 g during the same period. A fluctuating trend in TSS and titratable acidity was observed during the growth and development of bael fruit (Table 1) In CISH B-1, TSS remained almost constant up to 280 DAFS and then increased significantly from 21.5 to 31.8°B after 335 days of fruit set, which finally declined slightly to 29.3°B at 345 DAFS. Similar trend was noticed in CISH B-2 where it increased to 32.3°B at 335 DAFS and then decreased to 28.7°B at 345 DAFS. Titratable acidity increased significantly from 0.3% and 0.34% at 150 DAFS to 1.41% at 335 DAFS in both the selections and then decreased drastically to 0.46 and 0.51% in CISH B-1 and B-2, respectively, after 345 days of fruit set.
| Days after fruit set | Fruit weight (g) | TSS (°Brix) | Titratable acidity (%) | |||
|---|---|---|---|---|---|---|
| CISH B-1 | CISH B-2 | CISH B-1 | CISH B-2 | CISH B-1 | CISH B-2 | |
| 150 | 954 | 1,366 | 22.7 | 24.0 | 0.30 | 0.34 |
| 170 | 731 | 871 | 20.6 | 20.3 | 0.33 | 0.31 |
| 190 | 903 | 1,385 | 21.7 | 22.3 | 0.38 | 0.56 |
| 210 | 1,086 | 2,034 | 22.8 | 20.7 | 0.22 | 0.26 |
| 230 | 1,150 | 1,788 | 19.6 | 20.4 | 0.25 | 0.39 |
| 250 | 1,118 | 2,241 | 20.6 | 21.1 | 0.26 | 0.31 |
| 280 | 1,008 | 1,924 | 21.5 | 22.5 | 0.53 | 0.55 |
| 300 | 878 | 1,825 | 24.0 | 26.9 | 0.59 | 0.60 |
| 320 | 1,213 | 2,171 | 29.1 | 30.6 | 0.89 | 1.1 |
| 335 | 1,055 | 2,216 | 31.8 | 32.3 | 1.41 | 1.41 |
| 345 | 1,040 | 2,573 | 29.3 | 28.7 | 0.46 | 0.51 |
| CD at P<0.05 | 160.42 | 339.16 | 0.83 | 1.91 | 0.10 | 0.16 |
Table 1: Changes in average fruit weight, TSS and titratable acidity in bael selections during growth and development. Means of three replications (two fruits as one replicate).
Bael fruit is climacteric in nature. Fruit setting in bael takes place during first week of May and ripe fruits are available in the following April (approximately 11 months later) under central and north Indian conditions. Development in bael follows a single sigmoid curve. The growth rate of fruit has three distinct phases – the initial slow phase for one month (up to June) followed by a rapid increase up to September and then more or less a stationary phase up to harvest [15]. Fruits are generally harvested in a physiologically mature but unripe stage. It was observed that with the advancement of maturity fruit weight increased in both the bael selections. The fruit of CISH B-1 was 1.5 to 2 times smaller in weight than fruit of CISH B-2 throughout the developmental period. There was no significant difference in TSS, though slightly declining trend, observed in fruit of CISH B-1 and B-2 up to 280 DAFS, which significantly increased thereafter during maturity with slight decrease at the time of last harvest at 345 DAFS.
The literature on changes in TSS in bael during development is very scanty. However, in one bulletin it has been mentioned that TSS of CISH B-1 and B-2 pulp remained at 32 and 31°B at the time of harvest maturity [19]. Our results are in agreement with those found in literature for other fruits, where it has been reported that TSS content in guava fruit gradually increased with maturation except during the end of the growth period [20]. In mango, also TSS declined with the growth prior to physiological maturity and started rising till ripening [21]. Increase in TSS during fruit development and maturity has also been reported in case of guava [22] and cape-gooseberry [12]. Similar changing patterns were noticed in case of titratable acidity also, though the decline during final harvest was more pronounced. However, a continuous decrease in acidity in bael has been reported during development and maturity [1,19]. The increase in TSS and decrease in acidity during later period of development might be due to the formation of soluble and simple monosaccharides and disaccharides from complex and less soluble polysaccharides through hydrolysis, which indicated the onset of ripening in fruit.
Changes in AIS and total pectin
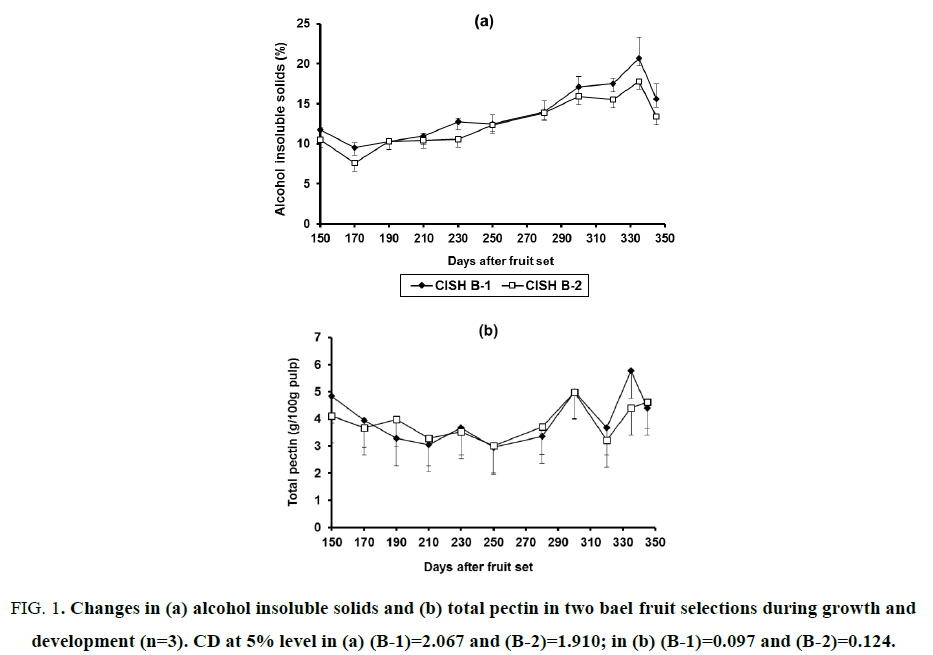
After an initial decline, AIS showed an increasing trend up to 335 DAFS and then decreased again in fruit harvested at 345 DAFS in both the selections (Figure. 1a). From 11.79 and 10.54% at 150 DAFS, AIS declined to 10.32% at 190 DAFS, thereafter increased to its peak level (20.7 and 17.8%) at 335 DAFS and finally decreased significantly to 15.6 and 13.4% at 345 DAFS in CISH B-1 and B-2, respectively. It did not show any specific trend during the development period. Fruit of CISH B-1 contained higher amount of AIS as compared to CISH B-2 throughout the growth period. The total pectin content showed a variable trend with less significant difference throughout the developmental period, though fluctuation in mean values was more as evidenced by error bars (Figure. 1b). When fruits were 150 days old, the contents of total pectin were 4.84 and 4.12% in CISH B-1 and B-2, respectively. In 345 days old fruits, total pectin contents were 4.40 and 4.62% for the said selections. It was noticed that the content of total pectin attained its peak level at 335 DAFS in fruit of CISH B-1 (5.77%) and at 300 DAFS in CISH B-2 (4.99%). In CISH B-1, the total pectin content was 5.07% at 300 DAFS. Similar to AIS, the amount of total pectin was also found higher in smaller sized fruit of CISH B-1 than in larger sized fruit of CISH B-2.
Figure 1: Changes in (a) alcohol insoluble solids and (b) total pectin in two bael fruit selections during growth and development (n=3). CD at 5% level in (a) (B-1)=2.067 and (B-2)=1.910; in (b) (B-1)=0.097 and (B-2)=0.124.
AIS in bael fruit (unknown variety) was reported to increase during development (with maturity) but decreased during ripening [1]. The rise in AIS content during development of mango fruit has been mentioned for cv. Dashehari [6]. An increase in AIS during growth of another mango cv. Alphonso has also been reported and the authors have stated that starch and other constituents contributed to the rise in AIS [23]. The decrease in AIS in two bael selections during initial stages of development might be due to the sharp decline in fruit weight at that period and the same during later stages of development might be due to the loss in hemicelluloses, celluloses, starch, etc. as bael fruit is rich in dietary fiber. The enzymatic de-esterification and hydrolysis of complex and insoluble carbohydrates to their soluble and simple forms might be another reason for sharp decline in AIS at 345 DAFS, which was also an indication for the onset of ripening in fruit. These observations were well supported in four guava cultivars where AIS declined gradually during the initial growing period of guava fruit followed by rapid decline during later stages of development [8]. The authors also opined that conversion of celluloses, hemicelluloses, pectin’s and starch to soluble constituents by some enzymes like pectinase, hemicellulase and cellulase might be the reason for decrease of AIS. The decrease in AIS with maturation has also been reported in blueberry fruit where increase in water uptake and subsequent increase in fruit size has been mentioned as the reason for the decline [13].
There were no significant changes observed in the amounts of total pectin during the development of bael fruit, though slight increase was noticed during later stages of development. This slight increase might be due to the increase in water-soluble fraction of pectin which constituted a major portion of pectin. Total pectin in this study was estimated by adding all the three fractions and not extracted from bael mesocarp. Because of the variations in three pectin fractions the significance of mean differences in total pectin was more as shown by error bars in (Figure. 1b). Singh and Roy [1] have observed an increase in pectin content during development of bael fruit. Our findings are analogous with those in mango [6], guava [22], cherry [11] and cape-gooseberry [12] fruits. With good amount of pectin, bael fruit can be used for the preparation of jam, jelly, marmalade, etc., if harvested at 300 DAFS.
Pectin transitions during growth and development
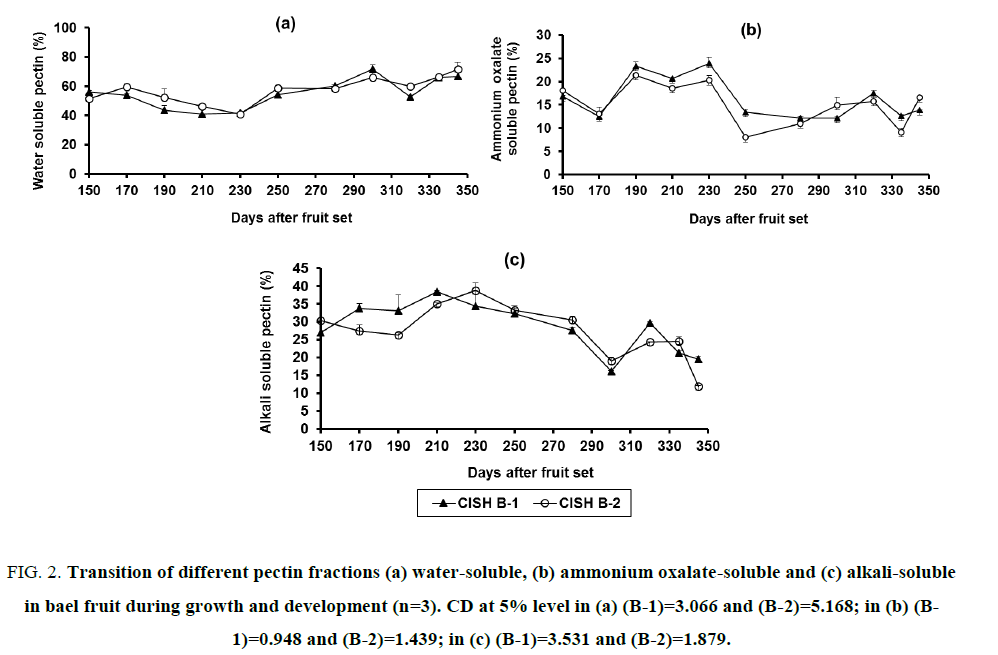
Upon characterization of AIS, the water-soluble pectin (WSP or pectinic acid with high methoxyl content) showed a sudden upward surge after 230 days of fruit set in both the bael selections. It increased thereafter with the advancement of fruit growth and reached maximum in CISH B-1 (71.62%) at 300 DAFS and in CISH B-2 (71.60%) at 345 DAFS (Figure. 2a). The content of WSP in CISH B-2 at 300 DAFS was 65.92%, which was slightly lower than the maximum amount. As the fruit approached maturity (335-345 DAFS), WSP content in CISH B-1 remained apparently constant but increased from 66.27 to 71.60% in CISH B-2. When fruits were 150 days old, WSP content was higher in CISH B-1 (56.31%) as compared to CISH B-2 (51.55%). However, in 345 days old fruit its concentration was significantly higher in CISH B-2 (71.60%) than in CISH B-1 (66.67%). The amount of ammonium oxalate-soluble pectin (OSP or pectic acid with low methoxyl level) remained low amongst the pectin fractions throughout the fruit development in both the bael selections. No specific pattern regarding changes in OSP during fruit development was observed (Figure. 2b), though the final concentrations (13.83 and 16.52% in CISH B-1 and B-2, respectively) were slightly less than that of initial concentrations (16.75 and 18.04% in CISH B-1 and B-2, respectively). The initial and final concentrations of OSP were slightly higher in CISH B-2 as compared to CISH B-1, but CISH B-1 contained higher OSP concentrations than CISH B-2 throughout the remaining growth periods. The highest amounts of OSP were recorded at 230 DAFS in both CISH B-1 and B-2, while the lowest amounts were found at 280 DAFS in CISH B-1 and at 250 DAFS in CISH B-2. Contrary to WSP, alkali-soluble pectin (ASP or protopectin) showed a continuous decreasing pattern during developmental stages in both the bael selections after showing an increasing trend during initial period of development (Figure. 2c). In CISH B-1, ASP increased from 26.94% at 150 DAFS to 38.41% at 210 DAFS and then declined gradually to 19.50% in fruits harvested after 345 days of fruit set. In CISH B-2, it increased from 30.41% at 150 DAFS to 38.70% at 230 DAFS and finally decreased to 11.88% after 345 days of fruit set. At 150 DAFS the content of ASP was higher in CISH B-2 than CISH B-1, but the trend was reversed when fruits were harvested after 345 days of fruit set. ASP attained its lowest concentrations at 300 DAFS in CISH B-1 (16.22%) and at 345 DAFS in CISH B-2 (11.88%). The content of ASP in CISH B-2 at 300 DAFS was 19.11%, which was second lowest.
Figure 2: Transition of different pectin fractions (a) water-soluble, (b) ammonium oxalate-soluble and (c) alkali-soluble in bael fruit during growth and development (n=3). CD at 5% level in (a) (B-1)=3.066 and (B-2)=5.168; in (b) (B-1)=0.948 and (B-2)=1.439; in (c) (B-1)=3.531 and (B-2)=1.879.
Though the significance of mean difference in all the three fractions of pectin is less, but when they are added to calculate total pectin the significance of mean difference is much higher as evidenced from both (Figure. 1b and 2b). There were no specific trends noticed in changes of water-, ammonium oxalate-and alkali-soluble fractions of pectin during growth and development of bael fruit, but at 345 DAFS WSP increased and ASP decreased significantly, while OSP declined slightly. This is more noticeable in bigger sized fruit of CISH B-2. The increase in WSP with fruit development might be due to the solubilization of pectic substances by pectic enzymes. During the development of mango, it was observed that WSP was maximal at harvest maturity, as in case of CISH B-2, and ASP was in peak few days earlier to the onset of maturity [6]. The increase in WSP and decrease in ASP contents were also reported by the same authors. Some authors have reported that ammonium oxalate-soluble pectin did not exhibit a regular pattern and the amount of WSP increased during maturation of mango [7], which is similar to our findings in bael. It has been mentioned in the literature that WSP content gradually increased with fruit development in guava cultivars [8]. The initiation of solubilization caused the increase in WSP content and decrease in ASP content during development of blueberry fruits [13]. An increase in water-soluble fraction and decrease in water-insoluble fraction of pectin during development and ripening of sweet cherry fruits have also been noticed [11]. The water-and oxalate-soluble pectic substances were found to increase, while those of acid-and alkali-soluble pectic substances decreased during development and ripening of cape-gooseberry fruits [12]. These authors also opined that the increase of WSP could be ascribed to solubilization of the esterified pectin by some enzymes. The hydrolysis of protopectin to water-soluble pectinic acid might cause the decline in ASP and rise in WSP during maturity and ripening of bael. The pectin fractions, especially WSP or high methoxyl pectinic acid and ASP or protopectin, play an important role during fruit maturation and ripening as evidenced from different studies. Different authors concluded that fruit attained maturity when WSP was at its peak and ASP was at its low. In the two bael selections, WSP was highest at 300 DAFS in CISH B-1 and at 345 DAFS in CISH B-2 while ASP was lowest at 300 DAFS in CISH B-1 and at 345 DAFS in CISH B-2. However, WSP and ASP were 65.92 and 19.11%, respectively, at 300 DAFS in CISH B-2, which were slightly less than the highest amount.
Conclusion
It was observed in the present study that with the advancement of growth and development in bael (irrespective of selections), AIS and WSP increased while ASP decreased. The transition of pectin fractions (increase in water-soluble pectin and decrease in alkali-soluble pectin) revealed that bael fruit could be harvested at 300 days after fruit set for processing purpose. In India, bael is generally harvested during March-April (300-335 DAFS) for use in processing industries. Total pectin content was also high during that period and bael can also be used as a good source of commercial pectin. Further elucidation on carbohydrate composition in isolated pectin is required to understand the ripening phenomenon in fruit.
References
- Singh RN, Roy SK. The Bael: Cultivation and processing. Indian Council of Agricultural Research, India. 1984.
- Maity P, Hansda D, Bandyopadhyay U, et al. Biological activities of crude extracts and chemical constituents of bael, Aeglemarmelos (L.) Corr, Indian J Exp Biol. 2009;47:849-61.
- Mohnen D. Pectin structure and biosynthesis. Current Opinions in Plant Biol. 2008;11:266-77.
- Thakur BR, Singh RK, Handa AK. Chemistry and uses of pectin:A review. Critical Rev Food SciNutr. 1997;37:47-73.
- Roe B, Bruemmer JH. Changes in pectic substances and enzymes during ripening and storage of ?Keitt? mangoes. J Food Sci.1981;46:186-9.
- Tandon DK, Kalra SK. Pectin changes during the development of mango fruit cv. Dashehari. J Hort Sci. 1984;59:283-6.
- Varanyanond W, Naohara J, Wongkrajang K, et al. Changes in pectin content and average molecular weight of pectin during maturation of the mango ?Kaew?. Food SciTechnol Res.1999;5:362-4.
- El-Buluk RE, Babiker EE, El-Tinay AH. Biochemical and physical changes in fruits of four guava cultivars during growth and development. Food Chem. 1995;54:279-82.
- Tiwari S, Esguerra EB, Dikshit A, et al. Pectin changes in specific gravity graded guava (Psidiumguajava L.) fruit cv. Sardar during low temperature storage at 5°C. Acta Hort. 2007;735:569-77.
- Georgiev Y, Ognyanov M, Yanakieva I, et al. Isolation, characterization and modification of citrus pectins. J Biosci Biotech. 2012;1:223-33.
- Fils-Lycaon B, Buret M. Loss of firmness and changes in pectic fractions during ripening and overripening of sweet cherry. Hort Sci. 1990;25:777-8.
- Majumder K, Mazumdar BC. Changes of pectic substances in developing fruits of cape-gooseberry (Physalisperuviana L.) in relation to the enzyme activity and evolution of ethylene. Scientia Horti.2002;96:91-101.
- Proctor A, Peng LC. Pectin transitions during blueberry fruit development and ripening. J Food Sci. 1989;54:385-7.
- Thomas M, Guillemin F, Guillon F, et al. Pectins in the fruits of Japanese quince (Chaenomeles japonica). CarbohydrPolym. 2003;53:361-72.
- Pandey D, Shukla SK, Pandey G. Bael (Aeglemarmelos Correa). In: Peter KV Underutilized horticultural crops. New India Publishing Agency. India. 2008;3:201-24.
- Ranganna S. Handbook of analysis and quality control for fruit and vegetable products. Tata McGraw Hill Publishing Company Ltd. India. 2000.
- Sheoran OP. OPSTAT Home Page http://www.hau.ernet.in/opstat.html. 2009.
- Roy SK, Singh RN. Studies on changes during development and ripening of bael fruit. Punjab Hort J 1980;20:190-97.
- Singh AK, Singh S, Singh RS, et al. The bael-fruit for dryland. Technical Bulletin No. 38 ICAR-Central Institute for Arid Horticulture. India. 2010.
- Rodriguez R, Agarwal PG, Saha K. Physico-chemical changes during development of safeda guava fruit. Indian Food Packer. 1971;25:5-20.
- Tandon DK, Kalra SK. Studies on developing mango fruits to assess maturity. Indian J Hort. 1986;43:51-9.
- El-Bulk RE, Babiker EE, El-Tinay AH. Changes in chemical composition of guava fruits during development and ripening. Food Chem. 1997;59:395-9.
- Lakshminarayana S, Subhadra MV, Subramanyam H. Some aspects of developmental physiology of mango fruit. J Hort Sci. 1970;45:133-42.