Research Paper
, Volume: 9( 4)Adsorption of copper (II) and cobalt (II) from model sewage onto modified bentonite.
- *Correspondence:
- A.Alemi
Institute of Chemical Problems Named afterAcademician M.F.Nagiyev of Azerbaijan National Academy of Sciences, (IRAN)
E-mail: aghz_bioch@yahoo.co.in
Abstract
Natural andNa-formof heat-treated bentonitewas applied for the adsorption of copper (II) and cobalt (II) ions frommodel sewage at 20-65°Ctemperature range. At various temperatures equilibrium adsorption capacity of both natural and Na-form heat-treated bentonite samples were determined. Adsorption efficiency of the adsorbents was increased by increasing the temperature. It can be hypothesized by the reduction of swelling and dehydration. Also, the adsorption efficiency for the Cu2+ was greater than Co2+ in all studied solution concentration and temperatures. Modification of bentonite by cation exchange method led into higher gross capacity values, equilibrium coefficient of distribution and metal ion adsorption selectivity.
Keywords
Adsorption; Modified bentonite; Copper; Cobalt; Bentonite.
Introduction
By rapid increase in global industrial activities, heavy metal pollution has become a serious concern. Due to the toxicity and non-biodegradable nature of heavy metals, the entry of them into water is becoming a critical environmental and public health field of attention [1-4]. Three kinds of heavy metals can be find in industrial wastewaters, including toxic metals (such as Hg, Cr, Pb, Zn, Cu, Ni, Cd, As, Co, Sn, etc.), precious metals (such as Pd, Pt, Ag, Au, Ru etc.) and radio-nuclides (such as U, Th, Ra, Am, etc.) [1,2]. According to this classification, metals such as copper, lead, zinc, cobalt, nickel, chromium and mercury have been considered as hazardous heavy metals and are very toxic elements which can be commonly found in water and wastewater. This fact can prove the necessity of the removal of these metals from wastewater [5,6].
Methods for metal ions removing from aqueous solution mainly consist of physical, chemical and biological processes [2]. The most important techniques include: precipitation [1], ion exchange [1,4], adsorption [1], coagulation [1,3], evaporation [7] and reverse osmosis [1]. Adsorption on solid matrices has been shown to be an economically feasible alternative method [3,6,8-10].
Copper is one of the most common toxic metals that find its way to water sources from various industries such as electroplating, mining, electrical and electronics, iron and steel production, non-ferrous metal industry, printing and photographic industries [6,8]. Trace amount (gL-1) of copper is required and necessary for some organisms as cofactors for the enzymatic activities. However, for the most of the organisms, concentrations at ppm (mg L-1) level are known to be toxic because of irreversible inhibition of some enzymes by the heavy metal ions [5,9-10]. According to U.S. Environmental Protection Agency (EPA) standards, the permissible limit of copper discharge in industrial effluents into water bodies is limited to 0.25 mg L-1 [6].
Cobalt containing compounds are widely used in many industrial applications such as mining, metallurgical, paints, pigment, and electronics. Cobalt is also present in the wastewater of nuclear power plants. The acceptable limits of cobalt in the irrigation water and livestock wastewater are 0.05 mg/L and 1.0 mg/L, respectively [5,11]. The acute cobalt poisoning in humans may cause serious health effects e.g. asthma like allergy, damage to the heart, causing heart failure, damage to the thyroid and liver. Cobalt may cause mutations (genetic changes) in living cells. Exposure to ionizing radiation is associated with an increased risk of developing cancer [5].
Activated carbon is a widely used adsorbent in the adsorption process, mainly due to its porous surface structure, which provides high surface area. The high cost of activated carbon operating has led to the development of new adsorbents with similar characteristics. On the other hand, recovering activated carbon particles from wastewater may be complicated [12]. Therefore, there is a need to explore alternatives to investigate low-cost, effective and economical adsorbents [5,11].
A low-cost adsorbent is defined as one which is abundant in nature, or is a by-product or waste material from another industry [5]. In addition to cost, adsorptive properties and availability are also criteria for choosing an adsorbent to remove pollutants. This has encouraged research into discovering materials that are both efficient and cheap [11]. In this manner, clay materials such as zeolite, montmorillonite, smectite, bentonite and sepiolite can be used as adsorbents for the adsorption process [5,10,13]. They are naturally occurring structured and phyllosilicate minerals, with high cation exchange and ion adsorption capacity [14].
Bentonite is a clay material (2:1 type), mostly contains montmorillonite. It is a soft, plastic and porous rock consisting mostly of calcium montmorillonite, with permanent negative charges on its [15]. The inner layer is composed of an octahedral sheet situated between two SiO4 tetrahedral sheets [10]. It is classified as a smectic soil composed of an expandable 2:1 type of alumino silicate clay mineral. It consists of flat particles with characteristic size of 102 to 103 nm with negatively charged surface ions and sodium counter-ions. The flat plates of clay have a tendency to pack parallel to each other forming liquid-crystalline order [9,11]. On account of higher surface area and cation exchange capacity, bentonite is a 2:1 mineral with one octahedral sheet and two silica sheets, which form a layer. Layers are held together by Vander Waals forces. Water can easily penetrate between layers of bentonite due to these weak forces, charge deficiencies and structure imbalances. Bentonite modification by cations could balance these deficiencies [14].
The charge imbalance is compensated by exchangeable cations including H+, Na+, or Ca2+ on the layer surfaces, in this way, modified bentonite is produced. The cation substitutions mainly take place in the octahedral sheets and may induce an enormous change in the physicochemical properties of clay minerals [5,13,16]. Chemical modifications on the surface of the bentonite with acids, bases, cationic surfactants and certain poly hydroxyl cations were also conducted to improve their adsorption capacity [11,15]. Bentonite has attracted great interest in environmental pollution management because of its outstanding properties such as high swelling ability and cation exchange capacity [15,16].
Al-Qunaibit [17] used bentonite for removal of high concentration of Cu (II) ions from aqueous solution. Zhao et al. [18] investigated the adsorption behavior of Cd(II) on GMZ bentonite. Zhi-rong and his coworker [8] studied the adsorption of copper and nickel on Na-montmorillonite as a function of solution pH, dosage of Na-bentonite, temperature and contact time. Xu et al. [19] surveyed the effect of shaking time, temperature, pH and ionic strength on the adsorption of Pb(II) onto MX-80 bentonite. They reported the adsorption thermodynamic parameters (i.e., ÄH0, ÄS0, ÄG0) and investigated the adsorption mechanisms of Pb (II) on the bentonite surface.
The aimof the presentwork is to studyadsorption of ions of copper (II) and cobalt (II) under static conditions onto natural andmodified bentonites, to performtheoretical calculations of some characteristics of adsorption process of the ions usingexperimental data.
Expermintal
Bentonite from Dash-Salahli deposit of Azerbaijan has the following characteristics: density of 2.22 g/cm3, SiO2:Al2O3 ratio about 5.7:2.1, composition of exchange cations, meq/100g sorbents – Na+(K+) 49.80; Ca2+(Mg2+) – 43.20, total capacity meq/100g sorbents – 93.00.
Adsorption experiments conducted in thermostatically controlled flask using of thermostat IT-1 up to 0.1°C in glass cells, where 0.05 g of adsorbent and 10 ml solution were contacted. Solution and adsorbent were mixed with glass stirrer at a constant rate. Concentrations of copper (II) and cobalt (II) ions in solutions were calculated by spectrometer on device SF-26.
Kinetic studies
Generalized rate constant of adsorption was calculated by ratio Sakovich [3]:

Where k is rate constant of adsorption process and n is a constant which depends on condition of experiment and mechanism of interaction of sorbing ion by adsorbent (0< n ≤ 1). Temperature dependences of rate constants of adsorption process of metal ions with bentonite containing adsorbent are well described by the equation of the following pattern:

Where, K is generalized rate constant of sorption, T is absolute temperature (K); C and d are constant, characterizing corresponding entropy and enthalpy of activation of adsorption process. Equation (3) is obtained for each dependence of equilibrium coefficients of distribution of metal ions Kd from equilibrium concentration of them in solution CT,∞ at different temperatures:
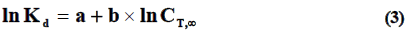
Where, Kd is equilibrium coefficient of distribution of metal ions in system (ml/g); a and b are constants for data of pH values of initial solution and temperature.
Johnson-Mehl-Avrami-Erofeev-Kolmogorov equation was applied to describe kinetics of adsorption of metal ions using Sakovich ratio [20,21]. Although this was developed by Avrami in the late 1930s, other workers were at the same time developing the same relationships and so the model is now referred to as the Johnson-Mehl-Avrami-Erofeev-Kolmogorov, or JMAEK, model. The JMAEK equation is [22]:
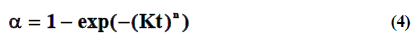
Where α is reached fraction, n is the Avrami exponent, K is the temperature-dependent rate constant.
The separation factor describes the selectivity of adsorbate between two metals ions in two systems. It is defined as the ratio of Kd for both ions as below, which A and B are different metal ions [23]:
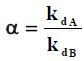
The thermodynamic parameters for the adsorption process ΔH (kJ/mol), ΔS (J/ (K mol)) and ΔG (kJ/mol) were evaluated using thermodynamic equations (5) and (6)
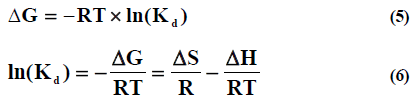
Where Kd is the adsorbate distribution coefficient;T is the temperature (K) and R is the universal gas constant (8.314×10-3 kJ/ (K mol)). Free energy ΔG0298.15 of copper (II) and cobalt (II) ions adsorption on different adsorbent changes correspondingly in the range of - 19.81 up to -26.85 by applying sewagemodel[24].
Results and Disscussion
One of the practically important tasks in environmental studies is modifying the surface characteristics of aluminosilicate adsorbents for increasing their tendency for inorganic compounds adsorption and improving technological properties of materials. Based on this fact, the heat-treated samples of aluminosilicate minerals were applied after modification in aqueous medium using chloride sodium salts, during adsorption experiments. A detailed description of obtaining method of ion exchange forms of aluminosilicate adsorbents was presented in the works [24]. The data of the experiments were performed in TABLES 1 and 2. The characteristics presented in TABLE 1 show the total capacity of bentonites with different chemical composition in the adsorption of sodium and calcium aluminosilicate minerals.
| Adsorbent | Cation of metal and pH solution | Adsorbent Properties | Adsorption of heat-treated bentonites at different temperatures | |||
|---|---|---|---|---|---|---|
| Temperature (1) 293.4 K |
Temperature (2) 308.4 K |
Temperature (3) 323.5 K |
Temperature (4) 338.5 K |
|||
| Na- Bentonite |
Cu2+ pH 7.8 |
Kd | 2.8·103 | 3.4.103 | 1.2·104 | 2.1·104 |
| CT, ∞ sorb. g/g | 29.72·10-3 | 31.2·10-3 | 34.5·10-3 | 35.1·10-3 | ||
| Natural Bentonite |
Cu2+ pH 7.8 |
Kd | 4.4·104 | 9.3·104 | 1.4·105 | 2.5·105 |
| CT, ∞ sorb. g/g | 21.24·10-3 | 24.6·10-3 | 28.01·10-3 | 31.2·10-3 | ||
| Na- Bentonite |
Co2+ pH 7.8 |
Kd | 1.3·103 | 4.2·103 | 1.4·104 | 3.0·104 |
| CT, ∞ sorb. g/g | 17.81·10-3 | 21.02·10-3 | 23.62·10-3 | 25.07·10-3 | ||
| Natural Bentonite |
Co2+ pH 7.8 |
Kd | 1.1·103 | 2.6·103 | 1.7·104 | 2.4·104 |
| CT, ∞ sorb. g/g | 12.42·10-3 | 17.16·10-3 | 20.02·10-3 | 22.04·10-3 | ||
| Na- Bentonite |
Cu2+ | Kd | 1.8·104 | 3.9·104 | 4.7·104 | 8.9·104 |
| CT, ∞ sorb. g/g | 20.4·10-3 | 22.7·10-3 | 26.8·10-3 | 29.1·10-3 | ||
| Na- Bentonite |
Co2+ | Kd | 0.86·103 | 3.9·103 | 8.4·103 | 2.2·104 |
| CT, ∞ sorb. g/g | 4.8·10-3 | 12.7·10-3 | 16.8·10-3 | 19.4·10-3 | ||
| Natural Bentonite |
Co2+ | Kd | 2.6·103 | 2.8·103 | 5.6·103 | 2.8·104 |
| CT, sorb. g/g | 7.8·10-3 | 12.7·10-3 | 15.01·10-3 | 17.9·10-3 | ||
| Natural Bentonite |
Co2+ | Kd | 1.1·103 | 1. 3 ·103 | 3.1·103 | 2.1·104 |
| CT, ∞ sorb. g/g | 6.91·10-3 | 8.34·10-3 | 11.7·10-3 | 14.8·10-3 | ||
TABLE 1: Calculated adsorption characteristics of natural and Na-forms of heat-treated bentonites during adsorption of copper (II) and cobalt (II) ions by using sewage model (size of adsorbent graining 2.5·10-2cm) Adsorption of heat-treated bentonites at different temperatures
| Temperature, K | Size of particles of Na-bentonite, cm | |
|---|---|---|
| 1.5-2.0·10-2 | 3.5-4.0·10-2 | |
| 293.4 | 8.25 | 8.70 |
| 308.4 | 8.52 | 8.45 |
| 323.5 | 9.38 | 9.29 |
| 338.5 | 9.91 | 9.83 |
TABLE 2: Values of logarithms of distribution coefficient (InKd) during adsorption of copper (II) ions from model sewage on heat-treated Na-bentonite with various sizes of graining
The replacement of Si by Na in bentonite structure (iso-morphous substitution) results in a deficit of positive charges or excess of negative charges, which are compensated by adsorption of a layer of cations that are too large to be accommodated in the different layers of structure. Na-bentonite is fully expandable, partially expandable, or non-expandable, depending on the surface charge density and the type and valance of the exchangeable cations. This cation that exists between the montmorillonite layers is an effective parameter for determining the characteristic properties of bentonite. Therefore, the diffusion coefficient of bentonite increases during surface modifications with cations such as Na+ [25,26].
Various bentonite structures have great potential to hold water. Interlayer water is lost at higher temperatures and the mesoporous character of the bentonite is preserved after the heating. As a result of these facts, the weight loss rate increased gradually with increasing temperature for bentonite. This weight loss is more tangible for Na-bentonite. Bentonite and modified bentonite structures have been both partially destroyed at high temperature, so this limitation must be considered in heat-treatments analysis [27,28].
As it is obvious, adsorption of Cu2+ and Co2+ ions using sewage model on Na–bentonite is higher than its natural analog due to mentioned reasons. The Na–bentonite adsorption capacity of Cu2+ and Co2+ ions is about 0.3–0.4 unit more than natural bentonite, which is Compatible with the literary data based on adsorption capacity of adsorbents [24]. Results given in the TABLE 1 and the calculations of gross capacity values of natural bentonite on copper (II) and cobalt (II) ions were compared with Na-bentonite. It is necessary to note that besides ion exchange adsorption, isomorphic replacement of Al3+ and Si4+ ions occur in octa-tetrahedral positions in crystalline lattice during adsorption processes of cations of transition metals. According to calculated values of free energy [24] and TABLE 1 data, it can be concluded that adsorption rate of metal ions changes according to: Cu2+ > Co2+. As it can be seen in TABLE 1, thermodynamic coefficients of copper (II) and cobalt (II) ions distribution, increased by raising the temperature treatment of bentonite. This enhancement is more obvious in Na-bentonite which can be explained by the mentioned reasons.
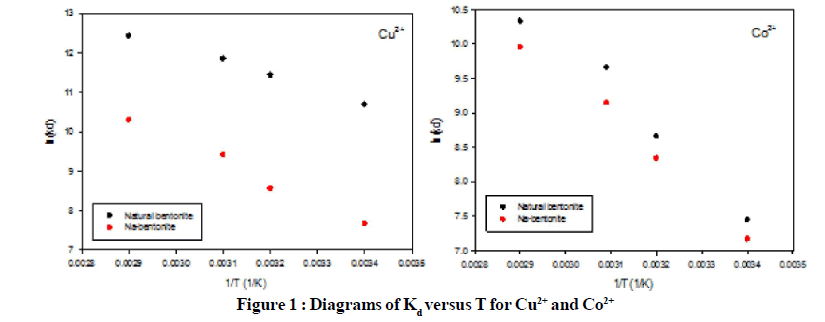
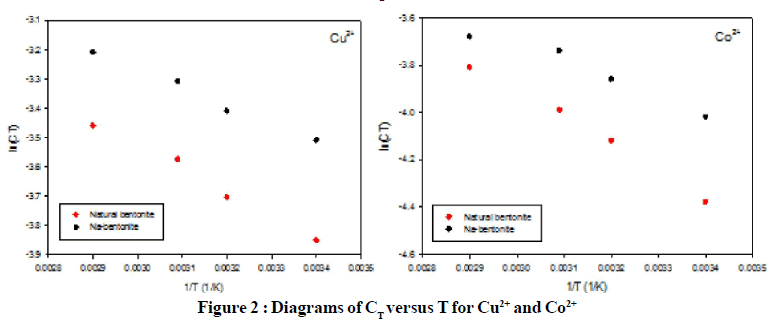
Figure 1 and Figure 2 show the CT and Kd variation with temperature for both Cu2+ and Co2+ adsorption on natural and modified bentonite. In Figure 1, lnKd of all cases decrease by decreasing the temperature because of reduction of moisture and swelling in the structure of both bentonite and Na-bentonite, while the slope of this reduction in Co2+ adsorption is more than Cu2+. The amount of Kd is higher for natural bentonite adsorptions. In Figure 2, lnCT in both diagrams decreased by temperature reduction. LnCT is higher in modified bentonite in all temperature for both Co2+ and Cu2+ adsorption which express the role of modification in enhancing the adsorption of heavy metal ions such as Co2+ and Cu2+.
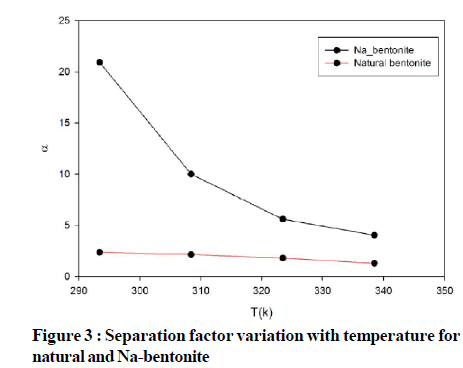
In two component adsorption systems with both Cu2+ and Co2+ ions, separation factor is defined for representing the selectivity of adsorbent regarding two ion metals. Figure 3 shows this selectivity of Cu2+ ions in comparing to Co2+ ions for both natural and modified bentonite. In both cases the separation factor decreased by increasing the temperature. As it can be seen, modification of bentonite increased selective adsorption of Cu2+ ions.
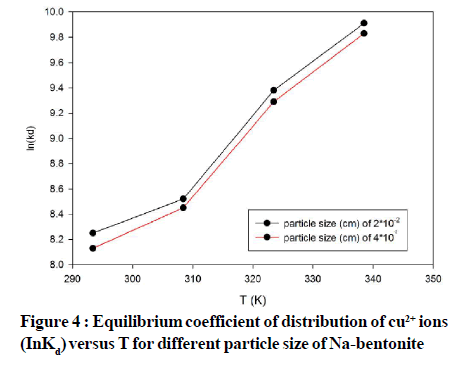
TABLE 2 shows the effect of Na bentonite particle size on Cu2+ ion adsorption. As it is seen in Figure 4 distribution coefficients of Cu2+ do not practically change during adsorption from model sewage with concentration of 6.32·10-5 mol/L at pH 7.8 with various sizes of particles within the limits of an error at a wide interval.
Figure 4: Equilibrium coefficient of distribution of cu2+ ions (InKd)versus T for different particle size of Na-bentonite
Conclusions
Adsorption of copper (II) and cobalt (II) ions using sewage model on natural and Na-form bentonites was investigated at 20-65°C temperature range with different Na particle size. Initial concentration of ions of copper (II) and cobalt (II) was 6.32·10-5 mol/L and 6.82·10-5 mol/L respectively. The equilibrium adsorption capacities of both natural and Na-form heat-treated bentonite samples at various temperatures were determined. Results indicated that the adsorption efficiency increased by increasing temperature. Also, the adsorption efficiency with the Cu2+ was greater than Co2+ in all studied solution concentration and temperatures. Static capacity of alumino-silicate adsorbents changes in the following order: Na-bentonite > natural bentonite. Results also revealed that granulated heat-treated Na-bentonite having sufficient mechanical strength and selectivity is found to be an efficient adsorbent for ions of Cu2+ and Co2+ and also can be successfully used for the extraction of ions of other transition metals from sewage in pseudo boiling layer. Finally it can be concluded that Na-bentonite particle size does not have significant effects on Cu2+ ion adsorption.
References
- F.Fu, Q.Wang; Removal of heavy metal ions from wastewaters:Areview. J.Environ.Manage, 92, 407-418 (2011).
- J.Wang, C.Chen; Biosorbents for heavy metals re-moval and their future. Biotechnol.Adv., 27, 195-226 (2009).
- B.A.M.Al-Rashdi, D.J.Johnson, N.Hilal; Removal of heavy metal ions by nanofiltration. Desalination, 315, 2-17 (2012).
- A.Dabrowski, Z.Hubicki, P.Podkoœcielny, E.Robens; Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method. Chemosphere, 56, 91-106 (2004).
- H.Ghassabzadeh, A. Mohaddespour, M.Ghannadi Maragheh, S.J.Ahmadi, P.Zaheri; Characterizations of Co (II) and Pb (II) removal process from aqueous solu-tions using expanded perlite. Desalination, 261, 73-79 (2010).
- N.Ertugay, Y.K.Bayhan; The removal of copper (II) ion by using mushroom biomass (Agaricus bisporus) and kinetic modelling. Desalination, 255, 137-142 (2010).
- A.Rether, M.Schuster; Selective separation and recovery of heavy metal ions using water-soluble N-benzoylthiourea modified PAMAM polymers. Reactive Funct.Polym., 57, 13-21 (2003).
- L.Zhi-Rong, Z.Shao-Qi; Adsorption of copper and nickel on Na-bentonite. Process Safety Environ.Protect, 88, 62-66 (2010).
- G.Zhao, H.Zhang, Q.Fan, X.Ren, J.Li, Y.Chen, X. Wang; Sorption of copper(II) onto super-adsor-bent of bentonite–polyacrylamide composites. J. Hazard.Mater, 173, 661-668 (2010).
- O.Gok, A.Özcan, B.Erdem, A.S.Özcan; Prediction of the kinetics, equilibrium and thermodynamic pa-rameters of adsorption of copper(II) ions onto 8-hydroxy quinoline immobilized bentonite. Colloids Surfaces A : Physicochem.Eng. Aspects, 317, 174-185 (2008).
- D.M.Manohar, B.F.Noeline, T.S.Anirudhan; Ad-sorption performance of Al-pillared bentonite clay for the removal of cobalt(II) from aqueous Phase. Appl.Clay Sci., 31, 194-206 (2006).
- H.Zheng, D.Liu, Y.Zheng, S.Liang, Z.Liu; Sorption isotherm and kinetic modeling of aniline on Cr-ben-tonite. J.Hazard.Mater., 167, 141-147 (2009) .
- B.Caglar, B.Afsin, A.Tabak, E.Eren; Characteriza-tion of the cation-exchanged bentonites by XRPD, ATR, DTA/TG analyses and BET measurement. Chem.Eng.J., 149, 242-248 (2009).
- A.L.Iskander, E.M.Khald, A.S.Sheta; Zinc and man-ganese sorption behavior by natural zeolite and ben-tonite. Annals Agric.Sci., 56, 43-48 (2011).
- M.Zamparas, A.Gianni, P.Stathi, Y.Deligiannakis, I. Zacharias; Removal of phosphate from natural waters using innovative modified bentonites. Appl.Clay Sci., 62-63, 101-106 (2012).
- A.Kul, H.Koyuncu; Adsorption of Pb(II) ions from aqueous solution by native and activated bentonite: Kinetic, equilibrium and thermodynamic study. J. Hazard.Mater., 179, 332-339 (2010).
- M.H.Al-Qunaibit, W.K.Mekhemer, A.A.Zaghloul; The adsorption of Cu(II) ions on bentonite-a kinetic study. J.Colloid Interface Sci., 283, 316-321 (2005).
- D.Zhao, S.Chen, S.Yang, Xi.Yang, S.Yang; Inves-tigation of the sorption behavior of Cd(II) on GMZ bentonite as affected by solution chemistry. Chem.Eng.J., 166, 1010-1016 (2011) .
- D.Xu, X.L.Tan, C.L.Chen, X.K.Wang; Adsorption of Pb(II) from aqueous solution to MX-80 bento-nite: Effect of pH, ionic strength, foreign ions and temperature. Appl.Clay Sci., 41, 37-46 (2008).
- J.Farjas, P.Roura; Modification of the Kolmogorov–Johnson–Mehl–Avrami rate equation for non-iso-thermal experiments and its analytical solution. Acta Materialia, 54, 5573-5579 (2006).
- A.Khawam, D.Flanagan; Solid-State Kinetic Mod-els: Basics and Mathematical Fundamentals. J.Phy.Chem.B, 110, 17315-17328 (2006).
- V.M.Gorbachev; Remarks on the Application of the Combined Kolmogorov- Erofeev- Kazeev- Avrami-Mampel Equation in the Kinetics of Non-isother-mal Transformations. J.Therm.Anal., 13, 509-514 (1978).
- F.W.E.Strelow; Quantitative separation of calcium frommagnesium, aluminum, irons(III) and many other elements by cation-exchange chromatogra-phy in methanolic hydrochloric acid on a macroporous resin. Anal.Chem.Acta, 127, 63-70 (1981).
- A.I.Yagubov, L.A.Binnatova, N.M.Muradova; Wastewater purification to remove dyes with monocation-substituted forms of bentonite and flocculants-coagulants. Russ.J.Appl.Chem., 83, 420-423 (2010).
- N.Güngör; Effect of the Adsorption of Surfactants on the Rheology of Na-bentonite Slurries. J.Appl.Polym.Sci., 75(1), 107-110 (2000).
- T.Melkior, E.C.Gaucher, C.Brouard, S.Yahiaoui, D. Thoby, Ch.Clinard, E.Ferrage, D.Guyonnet, C.Tournassat, D.Coelho; Na+ and HTO Diffusion in Compacted Bentonite: Effect of Surface Chem-istry and Related texture. J.Hydrol., 370(1), 9-20 (2009).
- Y.Sarikaya, M.Onal, B.Baran, T.Alemdaroglu; The Effect of Thermal Treatment on Some of the Physi-cochemical Properties of a Bentonite. Clays Clay Min., 48(5), 557-562 (2000).
- G.Yilmaz; The Effects of Temperature on the Char-acteristics of Kaolinite and Bentonite. Sci.Res.Essays, 6(9), 1928-1939 (2011).