Original Article
tsm, Volume: 12( 2)Controlling of Soil Born Plant Pathogen by Evaluating Fungal Biocontrol Natural Resources for Impartiality to Regularly Used Chemical Pesticides
- *Correspondence:
- Snehal F, Department of Microbiology, JB Science College, Nagpur University (MH), Wardha. India, Tel: 07152252528; E-mail: snehalfursule52@gmail.com
Received: May 12, 2017; Accepted: June 13, 2017; Published: June 19, 2017
Citation: Snehal F. Controlling of Soil Born Plant Pathogen by Evaluating Fungal Biocontrol Natural Resources for Impartiality to Regularly Used Chemical Pesticides. Macromol Ind J.2017;12(2):108.
Abstract
Background: The microbes which evolved from soil, have pathogenic in nature and their control is important procedure in the agricultural practise since it is based on the certain fungal biocontrol agent. So, isolation and identification of fungi by contaminated soil is the key role to evaluate natural resource by detecting the biocontrol activity of isolated fungi against plant pathogen. Meanwhile, here determine effect of pesticides on fungal biocontrol agent.
Aim: To determine the tolerance of fungal biocontrol resource to commonly used chemical pesticides for control of soil borne plant pathogen.
Methods: Soil sample is collected with history of repeated use of chemical pesticide then serially diluted the soil sample in a sterile distilled water. The soil sample is poured on the PDA plate. These plates are incubated for 24°C for 2 to 4 days. The fungal species which was identified and their characterization done by microscopic analysis based on morphological character. Then isolated fungus was followed by the detection of biocontrol activity.
Result: The biocontrol agents have been effectively work and help to reduce the soil born plant pathogen. The effect has been found only by mixing the application of biocontrol agents. It associates with the use of cheap fungicide like carbendazim, mancozeb which is commonly runs by farmers in their farms by low dose.
Keywords
Soil pathogen; Agricultural; Fungi; Chemical pesticides; Microscopic analysis; Biocontrol agents
Introduction
India is well known for the agricultural practices so the plant pathogen is the major thread now a days and framers are suffered by the problem. There is some scientist, who worked on the biocontrol agents of fungi and bacteria, which is associated with six pesticides that is frequently used in the control of soil borne plant pathogens. As the compatibility of fungal (Trichoderma harzianum, Trichoderma virens and Pochonia chlamydosporia) and bacterial biocontrol agents (Bacillus subtilis and Pseudomonas fluorescens) was used against six pesticides viz., carbendazim, mancozeb, metalaxyl, captan, thiram, and nemacur which is regularly used farmers in agricultural practices in India for the control of soil borne plant pathogens [1].
The isolation of various fungal strains is done by agricultural soil that is contaminated with pesticides. The occurrence of fungal strains from pesticides polluted agricultural soils that are categorized by twenty-three soil samples. The 59 fungal strains were isolated and 33 fungi were characterized using various isolation and identification methods. Soil samples were also characterized for physiochemical properties [2].
Methomyl and carbofuran degrading-bacteria from agricultural soils is isolated and classified in Split Valley and Central Kenya by the scientists. Moreover, they use of pesticides remains actual serious in defensive the farmers’ speculation in seeds, compost and labour since they provide a sure cover from damage by pests. The use of pesticides, environmental pollution due to pesticides and their residues will have continued to be a challenge. In this study, bacterial strains capable of degrading methomyl (S-methyl-N-[(methylcarbamoyl) oxy]-thioacetimidate) and carbofuran (2, 3-dihydro-2, 2-dimethyl-7-benzofuranyl methylcarbamate) was isolated [3].
Some of the suitable review of isolation, identification, and resistance profile with growth kinetics of chlorpyrifos resistant bacteria is given by some of the scientist by isolating it from agricultural soil. Although there are benefits to the use of pesticides, there are also drawbacks, such as potential toxicity to humans and other animals and change in the eco balance due to residual effects. Three isolates viz., Bacillus stearothermophilus, Bacillus circulans and Bacillus macerans were found resistant to 50 mg/L, 55 mg/L and 60 mg/L of chlorpyrifos [4].
Afield experiment was carried out during two consecutive years, 1999-2000 and 2000-2001 on clay-loam soils of Udaipur to examine the residual effect of incubated sources of Udaipur rock phosphate and FYM on soil in wheat-maize cropping sequence. Twenty six treatments were evaluated, 3 sources of Udaipur rock phosphate (URP 18%, 31% and 34%), 4 incubation methods (no incubation, incubation with phosphate solubilizing bacteria, incubation with farmyard manure and incubation with farmyard manure+phosphate solubilizing bacteria) and 2 farmyard manure levels (0 and 10 t ha) along with 2 checks (40 and 60 kg P O ha through DAP) [5].
Trichoderma spp has key role as to control of soil born plant pathogens which is used commonly in large amounts in agriculture for disease control and as well as to increased quality of the agricultural product [6].
One hundred and forty-six isolates of Trichoderma spp. were isolated in the middle areas of Gansu Province, China, from potato plants of rhizosphere soils. It has been identified by the dual culture technique only. They also examined antagonism against Fusarium sambucinum, which is related to potato dry root disease. From these ten of the isolates had found to be obviously antagonistic for that pathogen which based on morphological appearances with molecular analysis. The isolates have been identified viz Trichoderma harzianum Rifai, Trichoderma longibrachiatum Rifai, Trichoderma atroviride Karsten and Trichoderma virens which is antagonism. Among these isolates, D-3-1 (T. longibrachiatum) showed the strongest inhibition of the growth of Fusarium sambucinum [7,8].
Trichoderma species have been isolated from the soil only which is the part of plant root ecosystems. The fungi are not only resourceful but also help to fix nitrogen to plant. It is also helpful in reducing some of the diseases of the plant. As of now, we have been known to Trichoderma viride is the fungus species which is present in the soil. Meanwhile, is worked as potent Bio insecticide, Bio fungicide, Bio bactericide [9-11].
Trichoderma is used to prepare biofertilizer, found in all agricultural soils including forest humus layer and orchard soils. These free-living filamentous fungi are extremely interactive with root, soil and foliar environments. They are also very useful to make biofertilizer and helpful to increase the quality of the soil. It has also been worked against another microorganism which is harmful to agricultural field. The Trichoderma is the type fungi, which is association to control a various variety of plant pathogen fungi. It has several applications. It works antagonistic effect on plant pathogens. Meanwhile, it also works on positive effect on plant growth and yield in some vegetable crops [12,13].
By taking all above points in consideration, here the research aim is to determine the tolerance of fungal biocontrol agents to commonly used chemical pesticides for the control of soil borne plant pathogen. So that here will identified the biological natural resource which will stand for upcoming challenges for overcome the problem of soil borne plant pathogen.
Materials and Methods
Equipment
The equipment was used as Erlenmeyer flask, shaker, autoclave, petri plate, microscope, slide and forceps.
Media: Potato dextrose agar, sabouraud dextrose agar, CPZ agar.
Chemicals: Pesticides (carbendazin, mancozeb).
Reagents: Lacto phenol cotton blue.
Methodology
Soil sample collection: For the isolation of fungus or natural biocontrol resources, the soil sample were collected from the different area of farmer field where the history of repeated use of chemical pesticides.
Isolation of biocontrol agent/natural resources
1. The isolation done by the standard plating technique.
2. Primarily, the sol sample is serially diluted with sterile distilled water.
3. Then followed by the pouring on the potato dextrose agar plate.
4. Then followed by the Incubation on 24°C for 2 to 4 days.
5. After 2 days, the fungus colonies were observed on the PDA agar plate and composition shown in (Table 1).
| Ingredients | Grams |
|---|---|
| Potatoes (sliced washed unpeeled) | 200 g/liter |
| Dextrose | 20 g/liter |
| Agar powder | 2 g/liter |
| Water | 1000 ml |
Table 1. PDA composition.
Identification of fungi
The identification is done on the basis of the morphological characters. The other criteria are done by microscopic analysis, using taxonomic guides and standard procedure.
Staining method
Lactophenol cotton blue:
1. Take cotton blue (aniline blue) 0.05 g for 20 ml of dist. Water
2. Add phenol crystal 20 g.
3. Add glycerol 40 ml.
4. Mix with lactic acid 20 ml
5. Make up the volume with distilled water 20 ml.
The characterization was done by the staining technique:
1. The staining procedure required two days.
2. 1st day, take distilled water and dissolve the cotton blue in it, keep it for overnight to remove insoluble water.
3. 2nd day, take glass beaker; carefully add phenol crystal in lactic acid by wearing glows. Keep on magnetic stirrer until the phenol is liquefied.
4. Slowly add glycerol.
5. Filter the cotton blue and distilled water solution in to phenol/glycerol/lactic acid solution mix and store at 37°C.
Identification of the of the biocontrol agents based on morphological character by microscopic method:
1. Slide cleaning is most important here.
2. Take lacto phenol cotton blue of one drop.
3. Take thread like fungal species, spread on slide.
4. Put cover slip on to it.
5. Observe under the microscope.
Detection of bio control activity
The biocontrol activity was detected by the dual culture technique with various phytopathogen. In which the interaction/overlap zone between the antagonist fungus and plant pathogen is measured. In the petri dish PDA medium placed with 5 mm diameter disk with 5-day-old plant pathogen active mycelium and on the opposite site.
Trichoderma spp. is mycelium. The dishes were incubated at 28 ± 1°C and checked every 24 hr to count the number of days of the first contact between the antagonist and the phytopathogen; also, fungal growth of each colony was measured. In conclusion to, the diameter of the intersection zone was measured. Trichoderma overgrows the pathogen constantly and cover the culture medium surface completely. This classification done by Bell et al. (1982) where, Trichoderma overgrows two thirds of the culture medium; case 3, Trichoderma and the pathogen colonize approximately half of the surface of the culture medium and neither pathogen nor Trichoderma dominate; case 4, the pathogen colonizes at least 2/3 of the surface and resists Trichoderma invasion; case 5, the pathogen fungi colonizes the whole petri dish surface.
Observation and Result
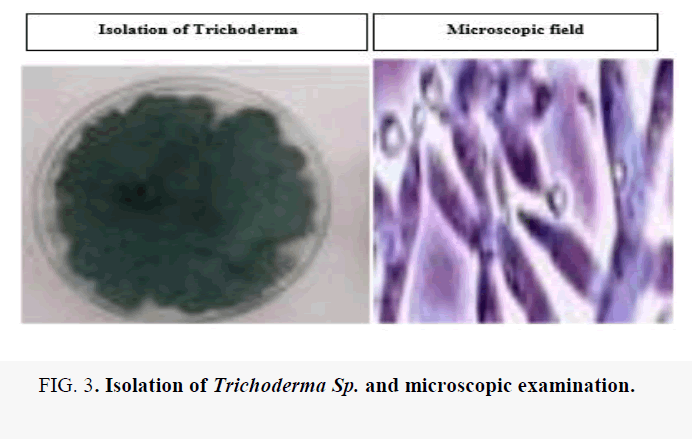
Isolation of Trichoderma spp. from the soil samples
Isolates of Trichoderma spp. were collected from Agricultural soils. After dilution technique some diluted sample is taken on the PDA at 28°C for 2 days, developed and became dark green at the center, shown in (Table 2).
| Biocontrol agent | Colour | Arrangement |
|---|---|---|
| T. harzianum | Dark green from the centre |
Pyramid shape, conidias spherical |
| T. longibranchiatum | Light green or deep green | Conidiophores oval |
| T. atroviridae | Dark green | Colonies spherical, conidiophores irregular |
| T.virens | Dark yellow from the centre | Conidia were oval |
Table 2. Identification of fungi.
Dual culture technique
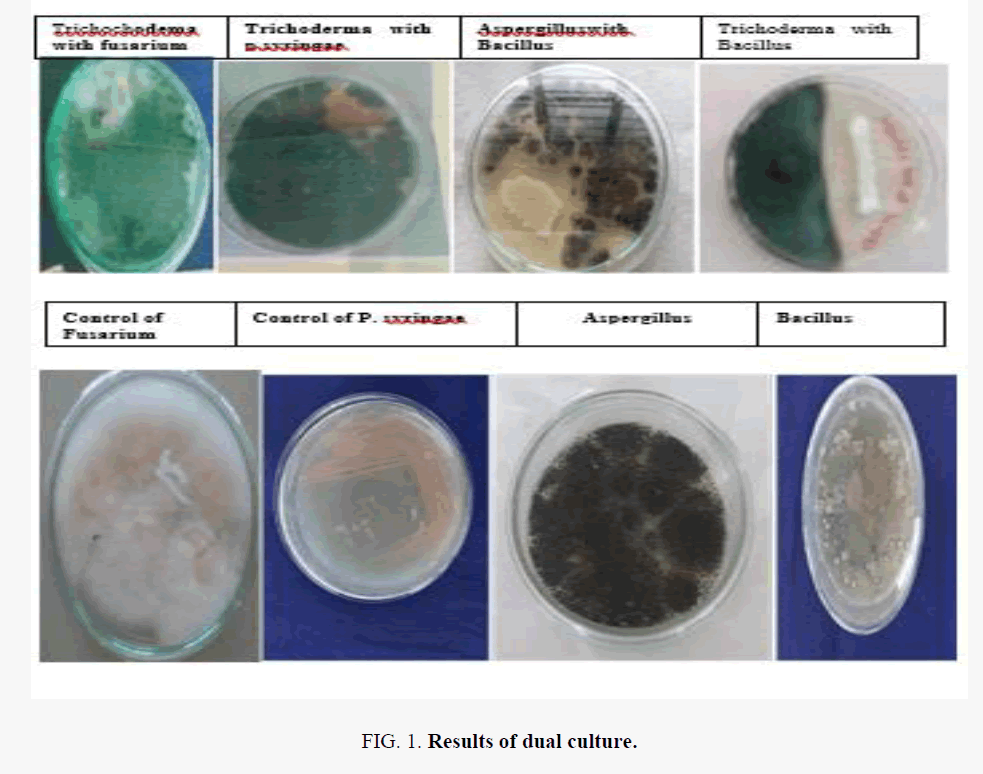
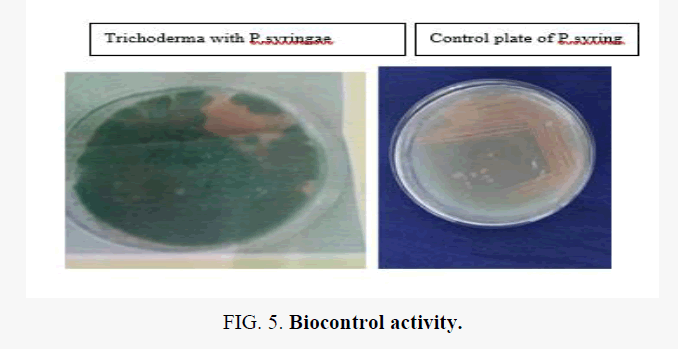
Results of dual culture assay (Figure 1), volatile metabolites and culture filtrate of Trichoderma spp. indicate that the tested Trichoderma sp. can inhibit the growth of fusarium, shown (Table 3).
| Name of the plant pathogen | Biocontrol activity |
|---|---|
| Trichoderma with fusarium | Positive |
| Trichoderma with P.syringae | Positive |
| A.Niger with Bacillus | Negative |
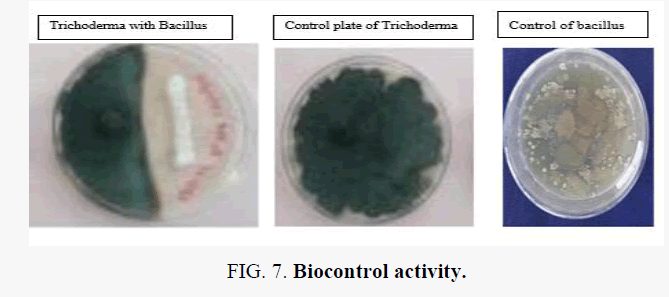
| Trichoderma with Bacillus | Positive |
Table 3. Effect of Trichodermal sp. against fusarium.
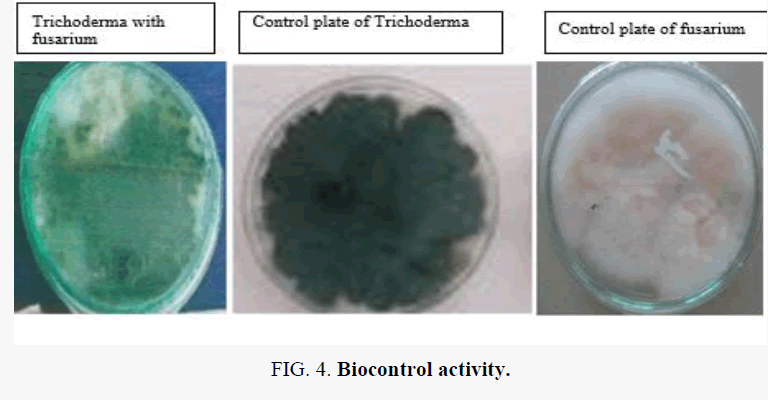
Trichoderma spp. can inhibit the growth of Fusarium. The results of dual culture showed that T. harzianum reduced the mycelia growth of Fusarium by 51.4% whereas T. viride inhibited the growth by 46.0%. Volatile metabolites from T. harzianum caused 38.1% growth inhibition of the pathogen while T. viride reduced the growth of Fusarium by 35.8%.
The culture filtrates of T. harzianum and T. viride caused appreciable reduction in the growth of Fusarium however none were fungicidal to the pathogen. Trichoderma sp. again can inhibit the growth of pseudomonas syringie. A. niger sp. can inhibit the growth of Bacillus. Trichoderma sp. again can capable of inhibiting the growth of bacillus, as shown in Figure 1.
Effect of pesticides
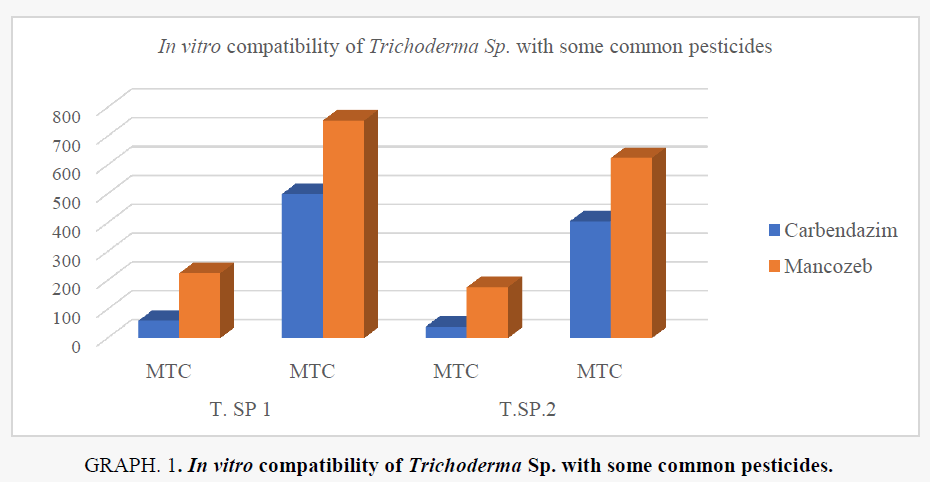
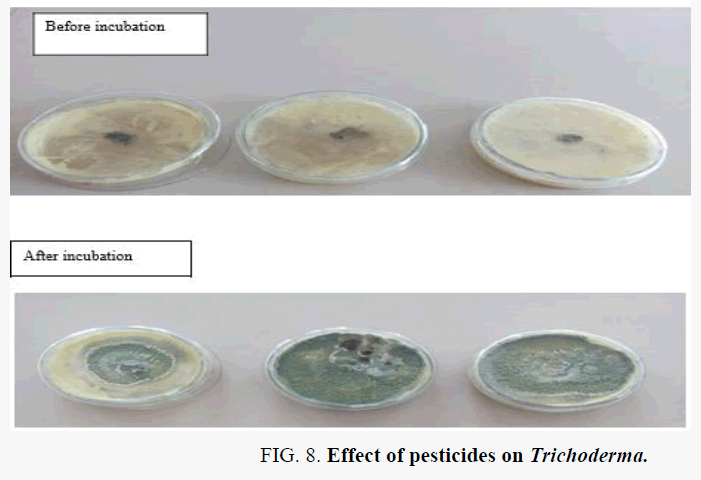
The maximum growth of T. harzianum was inhibited at a concentration of 500, 755, μg/ml of carbendazim, mancozeb, respectively. The corresponding value for T. virens were 405, 625 μg/ml of carbendazim, mancozeb, respectively. The fungicides at concentrations of 60 μg carbendazim/ml, 225 μg mancozeb/ml, for T. virens the safe tolerance limit was 40 μg carbendazim/ml, 177 μg mancozeb/ml showed less tolerance to the pesticides tested (Table 4). Biocontrol bacteria were found more tolerant to fungicides than fungi, shown in Figure 2 and Graph 1.
| Treatment | T. SP 1 | T.SP.2 | ||
|---|---|---|---|---|
| MTC | MTC | MTC | MTC | |
| Carbendazim | 60 | 500 | 40 | 405 |
| Mancozeb | 225 | 755 | 177 | 625 |
Table 4. Treatment of fungal species on common pesticide.s
Photo Gallery
The isolation of Trichoderma Sp. and microscopic examination for the morphological character is shown in Figure 3 and the biocontrol activity is shown in Figures 4-6 and Figure 7. The most important is effect of pesticides on Trichoderma shown in Figure 8.
Discussion
Here, the current study clearly established that, soil tolerated plant pathogens can be successfully managed by Trichoderma can parasitize fungal pathogens and yield antibiotics. Meanwhile, the fungus has many good application and has beneficial effects on plant growth, yield, nutrient uptake, increase fertilizer using efficiency, increase rate of seed germination as well increase the systemic resistance for plant diseases [14,15]. Cole and Zvenyika proposed that F. solani infection of tobacco plants could be controlled by incorporating T. harzianum into disease management procedures [16]. A study carried by Yedidia reported that T. harzianum inoculation improved the uptake of nutrients by the plant at a very early growth stage [17].
There is keeping in consideration, the relation between soil physicochemical belongings and richness of original microorganisms. The moisture content in soil acts as solvent and is essential for microbial functioning. A certain least level of natural matter and humidity content is essential to ensure the attendance of an active microbial population in the soil. In the present study, it is important to increase the agricultural yield pesticides plays a key role, but only less than 1% of the pesticides are enough to kill the pests, remaining pesticides enter the ground and surface water and causes environmental pollution [18]. Thus, some of the pesticides were banned and some are modified without damaging to the environment. However, organophosphates are creating a lot of health problems [19]. Various kinds of bacteria present in soil and water can degrade many insecticides. The present investigation involves the isolation of pollutants tolerant and resistant bacteria from the respective contaminated sites. As the microorganisms like bacteria plays a key role in bioremediation, experiment was carried out to identify the bacterial strain from organophosphate contaminated soil capable of degrading organophosphate. First, for the study soil samples were collected and it was serially diluted. Nutrient agar medium and PDA broth was prepared. The diluted soil samples are inoculated on to the PDA Petridis. An isolated colony was picked from the Petridis and later the same colony is inoculated onto the broth. From this pure, isolated and single colony were obtained after 4 to 6 times of subculture.
Here the physiochemical properties of soil keep in consideration for the study, used for isolation of microbial species. Color soil samples brown to black: in between pH (7.82-8.65). The temperature of the soil was high (30.2°C-33.2°C) with great variation in percent moisture content (0.35-0.95), organic carbon (0.2568-0.4125) and percent organic nitrogen (0.2213-0.3555). It come to know that Soil properties like organic matter, pH and moisture content etc., affects the density and diversity of microbes in the soil. Therefore, it is needed to study the relation between soil physicochemical belongings and plenty of indigenous microorganisms. The moisture content in soil acts as solvent and is essential for microbial functioning. A certain minimum level of organic matter and moisture content is essential to ensure the presence of an active microbial population in the soil. Here, the important physicochemical possessions of the soils, used for the valuation of natural fungal concentration, have been determined.
Conclusion
This study concludes is fungal strains were isolated from pesticide contaminated agricultural soil of different areas of the farm. These species successfully identified by the lactophenol cotton blue based stain and as well as characterized by the microscopic analysis with their morphology. Some of the fungal species that can grow efficiently, that’s means these original fungi have the capacity to adapt to xenobiotic compounds as novel growth and energy substrate. Therefore, these fungi have a potential to degrade xenobiotic compounds which will be tested in further study for degradation of commonly used pesticides.
Here, in this present study, it was clearly confirmed that, soil borne plant pathogens can be effectively accomplished by combined application of biocontrol agents/natural resources with inexpensive fungicide like carbendazim, mancozeb, and frequently used by Indian farmer at low doses. Also, pesticide contamination in soil will not affect the biocontrol agent effectiveness and hence can be easily applied in conjugation with the pesticides for the control of soil borne phytopathogen.
References
- Mohiddin FA, Khan MR. Tolerance of fungal and bacterial biocontrol agents to six pesticides commonly used in the control of soil borne plant pathogens. Afr J Agric Res. 2013;8:5331-4.
- Faryal RA, Hameed A. Isolation and characterization of various fungal strains from textile effluent for their use in bioremediation. Pak J Bot. 2005;37:1003.
- Omolo KM, Magoma G, Ngamau K, et al. Characterization of methomyl and carbofuran degrading-bacteria from soils of horticultural farms in Rift Valley and Central Kenya. Afr J Environ Sci Technol. 2012;6:104-14.
- Savitha K, Raman DS. Isolation, identification, resistance profile and growth kinetics of chlorpyrifos resistant bacteria from agricultural soil of Bangalore. Res Biotechnol. 2012;3.
- Rajput SS, Agarwal SK, Intodia SK. Residual effect of Udaipur rock phosphate sources, incubation methods and fym on soil fertility in wheat-maize sequence. Ind J Soil Conservation. 2014;42:164-9.
- Bokhari NA, Perveen K. In vitro inhibition potential of Phoenix dactylifera L. extracts on the growth of pathogenic fungi. J Medicinal Plants Res. 2012;6:1083-8.
- Nielsen KF, Gräfenhan T, Zafari D, et al. Trichothecene production by Trichoderma brevicompactum. J Agric Food Chem. 2005;53:8190-6.
- Samuels GJ, Dodd SL, Gams W, et al. Trichoderma species associated with the green mould epidemic of commercially grown Agaricus bisporus. Mycologia. 2002;94:146-70.
- Booker CJ, Bedmutha R, Vogel T, et al. Experimental investigations into the insecticidal, fungicidal, and bactericidal properties of pyrolysis bio-oil from tobacco leaves using a fluidized bed pilot plant. Industrial Eng Chem Res. 20101;49:10074-9.
- Singh HB, Singh BN, Singh SP, et al. Exploring different avenues of Trichoderma as a potent bio-fungicidal and plant growth promoting candidate-an overview. Rev Plant Pathol. 2012;5:315-426.
- Annadurai G, Rajesh Babu S, Mahesh KP, et al. Adsorption and bio-degradation of phenol by chitosan-immobilized Pseudomonas putida (NICM 2174). Bioprocess Biosyst Eng. 2000;22:493-501.
- Zaidi NW, Singh US. Trichodermain plant health management. Trichoderma: Biology and Applications. 2013:230.
- Agathos SN. " Biocatalytic weapons against micropollutants. Environ Eng Manag J. 2012;11:S99-104.
- Harman GE, Howell CR, Viterbo A, et al. Trichoderma species: Opportunistic, a virulent plant symbionts. Nat Rav Microbiol. 2004;2:43-56.
- Harman GE. Overview of mechanisms and uses of Trichoderma spp. Phytopathol. 2006;96:190-4.
- Gutierrez WA, Shew HD, Melton TA. Sources of inoculum and management for Rhizoctonia solani damping-off on tobacco transplants under greenhouse conditions. Plant disease. 1997;81:604-6.
- Sriram S, Manasa SB, Savitha MJ. Potential use of elicitors from Trichoderma in induced systemic resistance for the management of Phytophthora capsici in red pepper. J Biol Control. 2009;23(4):449-56.
- Kumari AR, Sobha K, Mounika K, et al. Molecular characterization of bacteria capable of organophosphate degradation. Int J Lifesc Bt & Pharm Res. 2012;1:64-72
- Yadav S, Verma SK, Chaudhary HS. Isolation and characterization of organophosphate pesticides degrading bacteria from contaminated agricultural soil. On Line J Biol Sci. 2015;15:113.