Original Article
, Volume: 11( 2)Green Synthesis of Gold Nanoparticles and Their Characterizations Using Plant Extract of Papaver somniferum
Wali Muhammad 1*, Sajjad AS1, Sumaira S2, Muhammad N3, Safia H1 and Muhammad J1
1Department of Biotechnology, Bacha Khan University Charsadda, KPK, Pakistan
2Department of Botany, Bacha Khan University Charsadda, KPK, Pakistan
3Department of Biotechnology, Malakand University Chakdarra, KPK, Pakistan
- *Corresponding Author:
- Muhammad W, Department of Biotechnology, Bacha Khan University Charsadda, KPK, Pakistan, Tel: +923455224994; E-mail: wali.biotech5511@gmail.com
Received Date: Jul 28, 2017; Accepted Date: Aug 30, 2017; Published Date: Sep 07, 2017
Citation: Wali M, Sajjad AS, Sumaira S, et al. Green Synthesis of Gold Nanoparticles and Their Characterizations Using Plant Extract of Papaver somniferum. Nano Sci Nano Technol. 2017;11(2):118
Abstract
Nanotechnology has a vast range of applications in medicines, electronics, biomaterials and energy production. The present research work is eco-friendly approach for the synthesis of gold nanoparticles using methanolic extract of Papaver somniferum. The appearance of deep purple color is the initial indication of gold nanoparticles synthesis. Papaver somniferum contains some active compounds which found to be responsible for the reduction of gold and convert it into a bioactive metal. The synthesized AuNPs were characterized by UV-Vis (UV-Vis spectrophotometer), SEM (Scanning electron microscopy), and FTIR (Fourier Transform Infrared Spectroscopy) FTIR. The UV spectra of gold nanoparticles showed absorbance band at 544 nm which is specific for gold. SEM analysis showed spherical morphology of synthesized AuNPs with average size of 77 nm. FTIR analysis showed the presence of various functional groups i.e. phenyl, ribose and amide which were responsible for the reduction and capping of AuNPs. The present work confirmed that Papaver somniferum already hold therapeutic promise and its conversion from extract to nanoparticles will be very helpful for medicinal application.
Keywords
Nanotechnology; Papaver somniferum; AuNPs; UV-Vis spectra; SEM; FTIR.
Introduction
Metallic nanoparticles have several applications like diagnostics, treatments and as a delivery system. Nanoparticles have distinct surface properties like larger surface area to volume ratio, and their modified properties as compared to bulk material [1,2]. Metallic nanoparticles display several size and shape-dependent optical properties, which are functional in several biomedical applications such as drug delivery, imaging of specific target cells and tissues, bio-sensing and catalysts to optics, etc. [3]. In specific, AuNPs have showed to be particularly resourceful for deep-tissue imaging. The perception of heavy metals can be positively included into clinical applications [4,5]. Physical methods assist a range of chemical methods to make AuNPs [6].
In such practices, traces of impurities stay in the gold colloidal solution showed the AuNPs incompatible for biological activity. As the living cells can reduce metallic ions to single valent form has led to the attention to make ‘green’ non-toxic reducing agents [7-10]. Green synthesis practices depend on biological materials like plants, algae and bacteria and are alternative of synthetic chemical reducing agents [11-13]. The green synthesis of AuNPs offers compensation counting amplified biocompatibility, easy to expand and reaction procedures. Several plant parts (stems, petals, bark, roots and leaves) can be subjugated as capping and stabilizing agents in the green synthesis of AuNPs [14,15]. Papaversomniferum (P. somniferum) belonging to the family papaveraceae. It is mainly cultivated in warm and temperate regions of the world [16].Papaversomniferum, commonly known as poppy plant [17]. Poppy have several medicinal properties and have been used for centuries in many ailments conditions [18-20]. Its seeds comprise analgesic potential and also used as source of oil [21-23]. The ethno-medicinal property of Papaversomniferum seed pulp extract incited us to carry out the present investigation to synthesize AuNPs in an eco-friendly manner.
Experimental Procedure
Materials required
Papaversomniferum plant pod part was homogenized via an electric blender to powder. About 5 g of the plants extract were incubated in dark for 2 days in 15ml methanol, later the extract was filtered via Whatman filter paper, the residue was then evaporated using a rotatory evaporator at 40°C. After cooling the sample was preserved in refrigerator for further analysis.
Synthesis of gold nanoparticles
Analytical grade gold chloride (HAuCl4) was procured from E. Merck. D-6100 Darmstedt, F.R. Germany. Stock solution 1 mM of HAuCl4 was prepared in distilled water. 700 ppm extract solution of each sample was prepared with 1 mM aqueous solution of gold chloride in different ratios by volume for synthesis of AuNPs were mixed and stirrer for 4 h till the color of solution completely change into yellow brown.
Characterizations
UV-Vis spectrophotometer
The reduction of gold ions was confirmed by measuring the UV-vis spectrum of the reaction mixture against distilled water ethanol mixture as a blank. The spectral analysis was done using UV-1602 Double Beam UV/Vis spectrophotometer BMS Biotechnology Medical Services at a resolution of 1 nm from 300nm to 800 nm. The selected ratio was freeze dry for further characterizations.
FT-IR spectroscopy
PerkinElmer spectrometer FT-IR SPECTRUM ONE in the range of 4000–0 cm-1 at a resolution of 4 cm-1 was used. The sample was mixed with KBr procured from Merck chemicals. Thin sample pellet was prepared by pressing with the hydraulic pellet press and subjected to FT-IR analysis. FTIR spectroscopy was carried out in department of Chemistry, Bacha Khan University Charsadda, KPK Pakistan.
Scanning electron microscopy (SEM)
Morphology of the sample was found out using FE-SEM (JSM-5910-JEOL-JAPAN). A pinch of dried sample was coated on a carbon tape. It was again coated with gold with auto fine coater (SPI-module sputter coater) and then the material was subjected to analysis.
Results and Discussion
Green synthesis of gold nanoparticles
The color of the reaction mixture following the addition of Papaversomniferum extract to the HAuCl4 solution altered from colorless to deep red when incubated in the dark. This color change designated the formation of NPs in the color arises due to excitation of surface plasmon vibration of AuNPs.
Optimization of gold nanoparticles
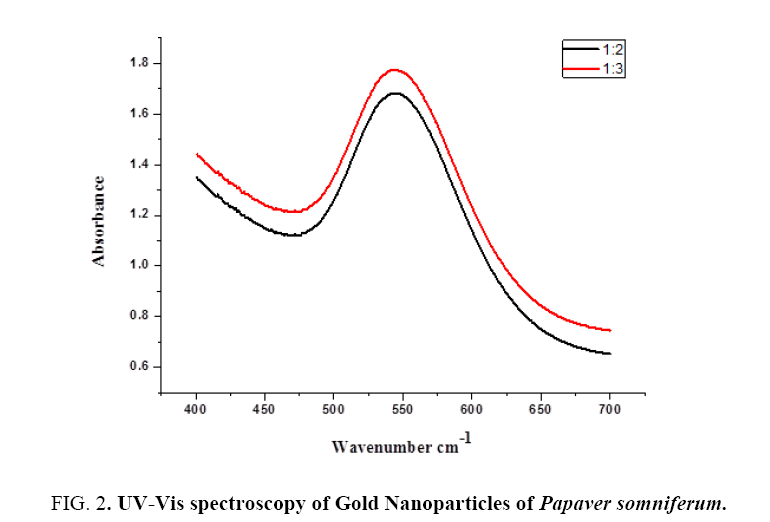
Different concentration is used for the optimization of AuNPs to confirm exact ratio of NPs synthesis which gives maximum absorbance at exact wavelength maximum value. We used two types of ratios 1:2 and 1:3. Both the ratios show absorbance bands in the range of 500 nmto 600 nm which is specific for AuNPs. 1:2 gives maximum absorbance value at exact lambda 544 nm. 1:3 also gives max absorbance value at exact lambda 543 nm (Figure 1).
Characterization results
UV-Vis spectroscopy: The Gold nanoparticles were visually established after the addition of Papaversomniferum extract into Gold solution. The variation in color from pale yellow into the deep red is the first sign of AuNPs synthesis. For additional conformation UV-Vis spectral technique is used. Various concentration of gold solution was subjected for optimization. Different ratios give different absorption peak but the most extreme peak with maximum absorbance was recorded 1:2 on 544 nm as shown in Figure 2. The wide spectrum showed that large size of gold particles formed due to Surface Plasmon Resonance (SPR). The UV-Vis analysis exposed that concentration of gold increase the frequency of particles decrease with increasing lambda max values. Our reports are also correlated with [24], by using aqueous extract of Calotropisprocera latex for the UV study and the UV spectrum showed absorbance band at 555nm. Our results are also similar with previous finding of [25], which carried out the green synthesis of gold nanoparticles by using extracts of Crocus sativus for the UV study and the UV spectrum showed absorbance band at 549nm.
Figure 2: UV-Vis spectroscopy of Gold Nanoparticles of Papaver somniferum.
Scanning electron microscopy: SEM is a sensitive technique in which electrons are used instead of light to form an image. So simply SEM is a type of electron microscope that produce images of a sample by scanning it with a focused beam of electrons. In the present research work scanning electron microscopy of AuNPs of extract of Papaversomniferumwas carried out to find the particles shape and morphology. The SEM results revealed that the gold nanoparticles possessed spherical shape with average particle size of about 77nm (Figure 3). Similarly, results were reported earlier for the gold nanoparticles like [26], carried out the biological synthesis of GNPs by using Magnolia kobusandDiopyros kaki leaf extract for SEM study and the images showed that the GNPs were triangular in structure with the size up to 300nm [25], carried out the green synthesis of gold nanoparticles by using Crocus sativus extracts for SEM study and the images indicated that the GNPs were spherical in nature with their size ranging from 11nmto 20nm.
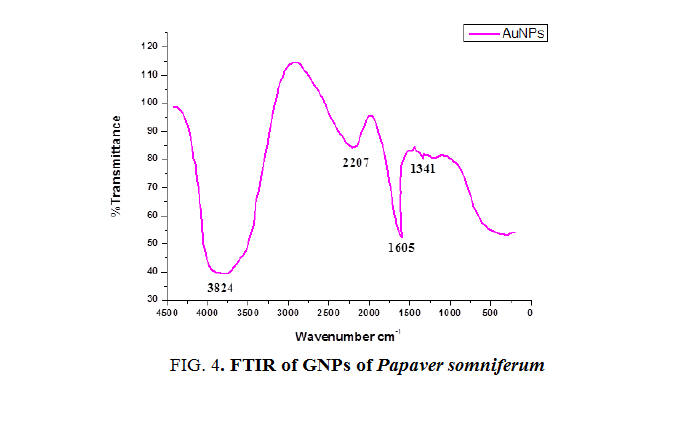
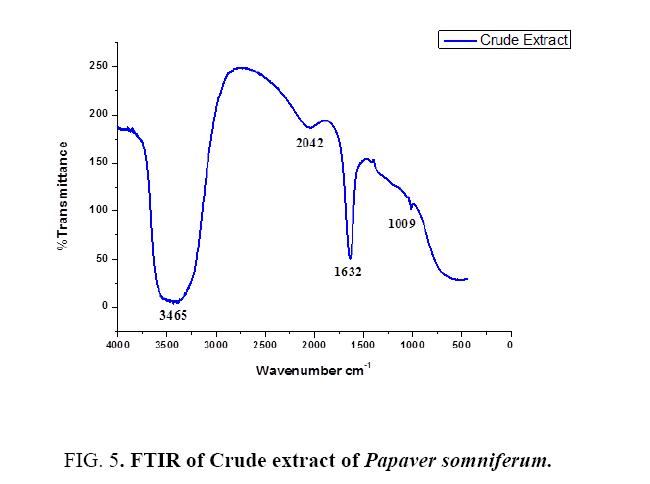
Fourier transform infrared spectroscopy: FTIR is used to obtain an infrared spectrum of absorption, emission and photoconductivity or Raman scattering of different materials. In FTIR spectrum the absorbance bands as shown in Figure 4 and 5 are observed in the region of 1000cm-1 to 4000 cm-1 of extract and AuNPs. FTIR analysis was performed to identify the possible biomolecules responsible for the reduction of Au+ ions and capping of the reduced AuNPs synthesized by using Papaversomniferummethanolic extract. The band at 1341 cm-1 in AuNPs corresponds to C-O stretching vibration with phenyl. The band at 1605 cm-1 corresponds to polysaccharides and pectin. The band at 2207 cm-1 corresponds to water molecule. The band at 3824 cm-1 corresponds to OH group. While in extract the band at 1009 cm-1 corresponds to ribose. The band at 1632 cm-1 corresponds to amide 1 and phenyl 1. Band at 2042 cm-1 corresponds to lipids and fatty acid and the band at 3465 cm-1 corresponds to OH bond. These bands are found in both and correspond to the different functional groups [27], carried out the green synthesis of GNPs using Cinnanomumzeylanicum leaf broth [28], carried out the green synthesis of GNPs by using Zingiber officinal extract, characterization and blood compatibility.
Conclusions
1. From current research work it is concluded that methanollic extract of P. somniferum having strong reduction efficacy of gold cations to gold nanoparticles.
2. Characterization of gold nanoparticles revealed that the average size of the synthesized nanoparticles is 77 nm with spherical in morphology and capped by phytochemicals.
3. Furthermore, it is recommended that the synthesized gold nanoparticles of Papaver somniferum can be used as catalysts and also for drug delivery and gene therapy in future studies.
Acknowledgment
The authors would like to thanks to Muhammad Haroon and Muhammad Azhar for providing plant material and helping with data analysis.
References
- Salata O. Applications of nanoparticles in biology and medicine. JNanobiotech. 2014;2:3.
- Rodriguez P, Plana D, Fermin DJ, et al. New insights into the catalytic activity of gold nanoparticles for CO oxidation in electrochemical media. Chinese J Catal. 2014;311:182-9.
- Cortie M, Van der Lingen E. Catalytic gold nano-particles. Mater Forum. Citeseer. 2002.
- Narayanan KB, Sakthivel N. Biological synthesis of metal nanoparticles by microbes.AdvColloid Interfac.2010;156:01-13.
- BoraU, Sett A, Singh D. Nucleic acid based biosensors for clinical applications. Biosen J. 2013.
- Babu P, SaranyaS, Sharma P, et al.Sonocatalyticsynthesis of gold nanoparticles using ethnolic extract of andrographispaniculata and functionalization with gelatin-polycaprolactone composites. Front Mater Sci. 2012;6:236-49.
- Patra S, Mukherjee S, Barui AK, et al. Green synthesis, characterization of gold and silver nanoparticles and their potential application for cancer therapeutics. Mater Sci Eng. 2015;53:298-309.
- Sheny DS, Philip D, Mathew J. Rapid green synthesis of palladium nanoparticles using the dried leaf of Anacardiumoccidentale. SpectrochimActa A. 2012;91:35-8.
- Iravani S. Green synthesis of metal nanoparticles using plants. Green Chem. 2011;13:2638-50.
- Mukherjee S, Sushma V, Patra S, et al. Green chemistry approach for the synthesis and stabilization of biocompatible gold nanoparticles and their potential applications in cancer therapy. Nano Technol. 2012;23:455103.
- Das RK, Gogoi N, Bora U. Green synthesis of gold nanoparticles using Nyctanthesarbortristis flower extract. Bioproc Biosynthesis. 2011;34:615-9.
- Agnihotri M, Joshi S, Kumar AR, et al. Biosynthesis of gold nanoparticles by the tropical marine yeast Yarrowialipolytica NCIM 3589. MatLet. 2009;63:1231-4.
- Gogoi N, Babu PJ, Mahanta C, et al. Green synthesis and characterization of silver nanoparticles using alcoholic flower extract of Nyctanthesarbortristis and in vitro investigation of their antibacterial and cytotoxic activities.Mater SciEng C. 2015;46:463-9.
- Awwad AM, Salem NM, Abdeen AO. Green synthesis of silver nanoparticles using carob leaf extract and its antibacterial activity. Int J Ind Chem. 2013;4:01-6.
- Babu PJ, Das RK, Kumar A, et al. Microwave-mediated synthesis of gold nanoparticles using coconut water. Int J Green Nanotech. 2011;3:13-21.
- Kirtikar KR, Basu BD.Indian medicinal plants. Indian Medicinal Plants. 1918:72.
- Evans WC.Trease and Evans' pharmacognosy, Elsevier Health Science. 2009.
- Kapoor L. Opium poppy:Botany, chemistry, and pharmacology. CRC Press. 1997.
- Barceloux DG. Medical toxicology of drug abuse: Synthesized chemicals and psychoactive plants. John Wiley Sons. 2012.
- A Century of International Drug Control, United Nations Publications. 2010.
- Robbers JE, Speedie MK, Tyler VE. Pharmacognosy and pharmacobiotechnology. Williams Wilkins. 1996.
- Chouvy PA. Afghanistan’s opium production in perspective. Chi EurFor Quart. 2006;4:21-4.
- Dewick P. Medicinal natural products: A biosynthetic approach. John Wiley Sons Inc. New York. 1997.
- Das RK, Sharma P, Nahar P, et al. Synthesis of gold nanoparticles using aqueous extract of Calotropisprocera latex. Mat Let.2011;65:610-3.
- Vijayakumar R, Devi V, Adavallan K, et al. Green synthesis and characterization of gold nanoparticles using extract of anti-tumor potent Crocus sativus.Physica E LowDimensSystNanostruct. 2011;44:665-71.
- Song JY, Jang HK, Kim BS. Biological synthesis of gold nanoparticles using Magnolia kobusand Diopyros kaki leaf extracts. ProcessBiochem. 2009;44:1133-8.
- Smitha S, Philip S, Gopchandran K. Green synthesis of gold nanoparticles using Cinnamomumzeylanicum leaf broth. SpectrochimActa A. 2009;74:3.
- Kumar KP, Sharma PW. Green synthesis of gold nanoparticles with Zingiberofficinale extract: Characterization and blood compatibility. ProcessBiochem. 2007;46:10.