Review
, Volume: 14( 4)Why Not Living Organisms or "Anomalous" Phenomena?
- *Correspondence:
- Carlos Eduardo Ramos CardosoDepartment of Meteorology, Federal Rural University of the Amazon, Belem, Para, Brazil; E-mail: carloseduardocardoso98@gmail.com
Received: September 12, 2024, Manuscript No. TSSE-24-147857; Editor Assigned: September 16, 2024, PreQC No. TSSE-24-147857 (PQ); Reviewed: September 30, 2024, QC No. TSSE-24-147857; Revised: August 06, 2025, Manuscript No. TSSE-24-147857 (R); Published: August 13, 2025, DOI. 10.37532/2319-9822.2025.14(4).394
Citation:Cardoso CER. Why Not Living Organisms or "Anomalous" Phenomena ? J Space Explor. 2025;14(4)394
Abstract
The study aims to scientifically reflect on the possibility of the existence or not of living beings or “abnormal” phenomena that go beyond our limited observations, considering the richness of elementary composition that performs a function such as protons, neutrons, electrons and elements of the periodic table related to the existence of living beings and phenomena. In addition, these elementary compositions are often used in the future for chemical reactions, that is, new functions that sustain living beings and phenomena on planet Earth. Therefore, based on a limited reference concentration of exploration of the universe, a new composition that goes beyond our limited reference is possible and this composition can possibly be used to carry out chemical reactions providing new functions that sustain the existence of living beings or abnormal phenomena.
Keywords
Abnormal life; Abnormal phenomena; Limited reference; Composition and reaction
Introduction
There is an intense attempt to know the universe beyond planet Earth. This attempt strongly uses the events on planet Earth as a reference to know the universe. An example is the search for life outside of Earth using the life currently on planet Earth as a reference. In other words, many scientists argue the need for specific factors (liquid water, adequate temperature, atmosphere, among others) that can help in the existence of life. This practice of searching for life based on these specific factors is coherent, but it is necessary to observe the variety of elemental composition and chemical reaction on planet Earth that implies the new element or fact, that is, it is capable of expressing functions and these chemical elements that express such characteristics sustain different living systems (plants, animals and bacteria).
In this sense, if there are new elemental compositions outside of Earth? If this composition has the ability to make chemical reactions? Will it be possible to sustain new living systems or new “abnormal” phenomena? It is incorrect to understand the elements in a final way. In other words, it is necessary to understand the behavior and the development process of the element [1].
Furthermore, it should be noted that limited referential concentration may be related to error, general accuracy or partial accuracy [2].
Literature Review
Variety of particle compositions
The reality of a variety of particles known to humans shows that there is not just one case of particles, making it possible for new particles to exist outside our galaxy. Furthermore, it is necessary to highlight the numerous differences in the characteristics that enable functions such as the proton that helps determine an element, the electron in electricity and the neutrons in the stability of the atom. Currently, there are numerous particles known to the scientific community. Thus, it is observed that new particles can be found outside our galaxy that support abnormal elements or facts [3].
The proton is composed of three quarks which are two up quarks (u) and one down quark (d) and have a positive charge. Electrons are fundamental subatomic particles and, unlike protons and neutrons, are not composed of smaller particles. They are considered elementary particles and have a negative charge [4].
A neutron is composed of three quarks: two down quarks (d) and one up quark (u) and has a neutral charge. Furthermore, it is important to remember that there are other varieties of particles that perform a function, showing that there is not just one case of scientifically possible composition.
Elementary compositions in the periodic table
Therefore, new or slightly different particles can support new elements similar to the known periodic table, and can be used for new chemical reactions that support abnormal phenomena or living beings [5].
The periodic table that organizes the chemical elements occurs due to the elementary composition of protons being an important factor in defining the chemical element.
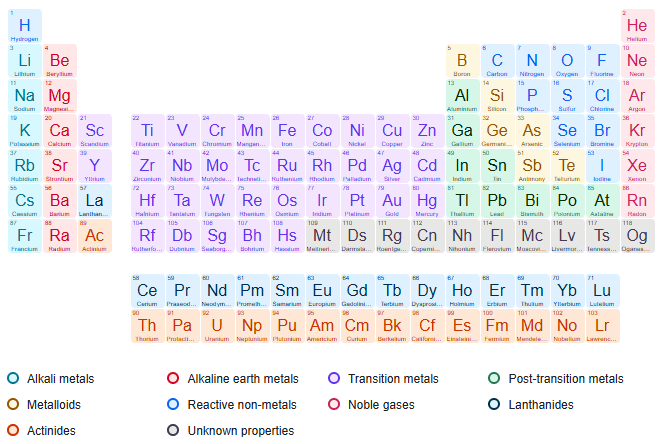
Furthermore, it is possible to observe that this elementary composition provides elements with physical concepts different from other chemical elements in addition to being used in future chemical reactions (Figure 1).
FIG. 1. Periodical table.
In this sense, we observe a variety of chemical elements that perform numerous functions and utilities, through the composition of protons. Thus, based on a variety of scientific discoveries of particles, it is understandable that the behavior and formation process of particles is varied, that is, a new particle that provides functions and utilities equal to the proton is possible. Thus, it is necessary to demonstrate the variety of functions and utilities of chemical elements in the periodic table. There are currently 118 chemical elements, which are in the table in increasing order of atomic numbers from left to right [6].
Examples of groups in the periodic table:
It is possible to observe the variety of some characteristics between groups in the periodic table.
Metals: Metals are the most common elements in the periodic table. Some examples are gold, silver, copper, zinc, iron, platinum, aluminum, sodium, potassium, among others. The elements belonging to this group have the following main properties:
- To shine
- They are solid
- Conducts electric current
- Conducts heat
- They are malleable
- They are ductile
Non-metals: They are composed of 11 elements carbon, nitrogen, phosphorus, oxygen, sulfur, selenium, fluorine, chlorine, bromine, iodine and astatine that have different properties from metals:
- Don't shine
- Does not conduct electricity
- Does not conduct heat
- Fragmentation occurs
Semimetals: They are composed of 7 elements boron, silicon, germanium, arsenic, antimony, tellurium and polonium that have intermediate properties to metals and non-metals:
- They have shine
- Poor conduction of electricity
Noble gases: These are the elements of family 18 of the periodic table. They are helium, neon, argon, krypton, xenon and radon.
Hydrogen: Hydrogen is different from any other chemical element, as it does not fit into any of the groups presented.
Periodic table family examples
Furthermore, it is possible to be more specific in demonstrating the variety of characteristics using families from the periodic table.
Family of alkali metals: They are metallic solids, shiny, soft, high thermal conductivity, high electrical conductivity, relatively low melting temperatures and are highly reactive with water.
Family of alkaline Earth metals: They are metallic solids, shiny, hard, they oxidize quickly, they have high thermal conductivity, high electrical conductivity, they have slightly higher melting temperatures compared to alkali metals and they are reactive with water.
Family of transition metals: Hard, shiny metallic solids, high thermal conductivity, high electrical conductivity, dense and high melting temperatures.
Boron family: They have intermediate properties between those of metals and non-metals and are solid under ambient conditions.
Carbon family: They have intermediate properties between the properties of metals and non-metals, they are solid under room conditions, they tend to form four bonds and they are solid under room conditions.
Nitrogen family: They have intermediate properties between those of metals and non-metals and are solids under ambient conditions, with the exception of nitrogen, which is a gas.
Oxygen family (chalcogens): They have varied properties, changing from non-metallic to metallic elements as one moves down the family under ambient conditions, with the exception of oxygen, which is a gas.
Halogen family: They are non-metals, quite reactive, poor thermal and electrical conductors.
Family of noble gases: They are non-metals, not very reactive and exist in the form of gases.
Discussion
Examples of specific elements from the periodic table
Potassium is a chemical element of the alkali metal group: Solid at room temperature, melting point is 63.5°C, boiling point is 759°C, Density is 0.89 g/cm3, extremely reactive, exothermic, oxidizes quickly, electronegativity is low, forms ionic compounds such as Potassium Chloride (KCl) and Potassium Nitrate (KNO3). It is not found in elemental form in nature due to its reactivity.
Essential for cellular function, especially in maintaining membrane potential and conducting nerve impulses. It is one of the main intracellular ions in living organisms. Used in fertilizers due to its important role in plant nutrition. It is also used in the manufacture of soaps.
Magnesium is a chemical element of the alkaline Earth metals group: Solid at room temperature, melting point is 650°C, boiling point is 1,090°C, density is 1.738 g/cm³, reactive, forms a protective oxide layer when exposed to air, which helps prevent corrosion, Relatively low electronegativity, Forms ionic compounds such as Magnesium Chloride (MgCl2) and Magnesium Sulfate (MgSO4).
Magnesium is the eighth most abundant element in the Earth's crust and is found in minerals such as Dolomite (CaMg(CO3)2) and Magnesite (MgCO3). It is also present in large quantities in seawater.
Essential for living organisms. It is a cofactor in many biological processes, including photosynthesis, where it is present in the center of the chlorophyll molecule. It also plays an important role in muscle function and bone stability.
Used in the manufacture of light alloys for automotive and aerospace components. It is also used in the production of fireworks, flares, and in metallurgical processes.
Gold is a chemical element that belongs to the group of transition metals: Solid at room temperature, melting point is 1,064°C, boiling point is 2,856°C, density is 19.32 g/cm3, excellent conductor of electricity and heat, very stable, resistant to corrosion and oxidation, electronegativity is relatively high for a metal, forms few compounds and does not react easily with most acids, but dissolves in aqua regia (a mixture of hydrochloric and nitric acid), forming Gold Chloride (AuCl3).
Aluminum is a chemical element that belongs to group 13 of the periodic table, in the boron family: Solid at room temperature, melting point is 660.3°C, boiling point is 2,470°C, density is 2.70 g/cm3, good thermal and electrical conductivity, quite reactive, electronegativity is moderate, forms a variety of compounds, such as Aluminum Oxide (Al2O3), Aluminum Chloride (AlCl3) and Aluminum Sulfate (Al2(SO4)3).
It is found mainly in minerals such as bauxite, which is the main source of aluminum in industry. Widely used in the manufacture of light alloys for the aeronautical and automotive industries, packaging and civil construction.
Lead is a chemical element of group 14 of the periodic table: At room temperature, melting point is 327.5°C, boiling point is 1,749°C, density is 11.34 g/cm3, conducts electricity and heat, resistant to corrosion and reactivity, forms several compounds such as Lead Oxide (PbO), Lead Dioxide (PbO2), and lead salts such as Lead Acetate (Pb(C2H3O2)2) and Lead Nitrate (Pb(NO3)2), highly toxic to humans and animals.
Lead is found in the Earth's crust mainly in minerals such as galena (PbS), which is the main source of lead extraction.
Lead is used in a variety of industries, including the manufacture of lead-acid batteries, solders, anti-corrosive coatings, radiation shielding, and metal alloys. It was also widely used in paints and plumbing, although this use has declined due to concerns about toxicity.
Nitrogen is a chemical element belonging to group 15 of the periodic table: Gas at room temperature, melting point is -210.1°C, boiling point is -195.8°C, density is 1.2506 g/L, forms a variety of important compounds including Ammonia (NH3), Nitrogen Oxides (NO, NO2), Nitric Acid (HNO3) and Nitrates (NO3-) and low solubility in water compared to other gases.
Therefore, based on countless examples of the variety of characteristics of the elements in which the composition differentiates them, it is possible to deduce the power of such a composition to promote such characteristics. Furthermore, it is necessary to understand the scientific reasons that make the element differentiable from the others.
Chemical reactions for new functions
The compositions of the particles that form a composition of atoms in the periodic table can be used to carry out chemical reactions, enabling new functions that sustain living beings or phenomena on Earth.
Example of cellular respiration
Cellular respiration is the process by which organisms obtain energy to carry out a wide range of activities. In cellular respiration, energy is obtained by oxidizing a glucose molecule, releasing energy. Part of this energy is stored in the form of ATP (Adenosine Triphosphate) molecules, and part is released as heat.
This process can be divided into three stages: glycolysis, the citric acid cycle and oxidative phosphorylation. In general, the term cellular respiration is used by scientists to represent phases two and three processes that occur in mitochondria.
formula in question is:
C6H12O6+6O2 → 6CO2+6H2O+Energia
This reaction is essential for energy production in aerobic organisms.
Example of photosynthesis
Photosynthesis is a process by which solar energy is transformed into chemical energy to synthesize organic compounds. Photosynthesis is primarily responsible for energy production in the biosphere and is carried out by plants or algae. It is an essential process for maintaining life on Earth, playing a fundamental role in energy production, oxygen release and regulation of the carbon cycle.
Water example
Water is a substance formed by two hydrogen atoms and one oxygen atom (H2O).
These elements are joined together by bonds called covalent bonds, which are characterized by the sharing of electrons present in the last electronic layer to obtain stability
In living beings, water performs various functions: Transport of substances throughout the body, elimination of toxic or excess substances, thermal regulation of the organism and composition of cells, tissues and organs. The human body has about 70% composed of water.
Water can exist in three main physical states: solid, liquid, and gas. Each state is characterized by specific properties and behaviors.
Solid state:
Water temperature turns to ice at 0°C under normal atmospheric pressure.
Structure of water molecules in the solid state form an organized crystal lattice structure, where hydrogen bonds hold the molecules in a fixed configuration.
Properties of the solid is less dense than liquid water, which causes it to float. In addition, ice is solid and has a defined shape.
Liquid state:
Water temperature is in the liquid state between 0°C and 100°C under normal atmospheric pressure.
In the liquid state structure, water molecules are in constant motion and do not have a fixed structure. Hydrogen bonds still exist, but they are more flexible and transient.
Properties of liquid water It has a shape that adapts to the container that contains it. It has high cohesion and adhesion, and its density is maximum at about 4°C.
Gaseous state:
Water turns to vapor at 100°C at sea level, although the transition to the gaseous state can occur at lower temperatures under reduced pressure.
In the gaseous state structure, water molecules are very separate and move freely, without significant interactions between them. Properties of gas has no defined shape or volume and expands to fill any available space. The density of vapor is much lower than that of liquid or solid water. In this way, chemical reactions have processes that sustain life on Earth.
limited reference concentration
It is essential to understand that a limited referential concentration may be related to scientific accuracy, but it may also be related to scientific error, given that a referenced concentration does not satisfy the whole. In this sense, other ways of thinking are needed to achieve the desired scientific accuracy that goes beyond understanding the final element or phenomenon, that is, it is necessary to understand the reasons and processes that promote these elements or phenomena. Thus, any understanding deemed correct based on the limited referential concentration about the origin of the universe or its evolution may be related to error or accuracy.
Imagine a person who does not know planet Earth and has never left the Amazon in Brazil. This person wants to understand planet Earth only with limited, concentrated observation of the Amazon. Therefore, the person comes to the wrong conclusion that the entire planet Earth is made up of trees. Furthermore, it is possible to observe through the variety of elements (which have the power to produce phenomena) on numerous planets that the distribution does not always occur in the same way, showing that the information contained is only in that space and cannot be generalized. Therefore, what is the safe guarantee that the universe evolved distributing elements or phenomena in the same way as observed?
It is worth noting that NASA's James Webb Space Telescope can only observe a part of the universe, that is, the whole is not observed. Thus, it characterizes a limited concentrated observation, and perhaps the studies developed may represent only a part that is observed.
Conclusion
Therefore, based on the elemental compositions that promote life-sustaining characteristics and phenomena, these compositions can be used in the future for chemical reactions that also promote more life-sustaining characteristics and phenomena. Thus, it is necessary to understand the reasons that promote these distinguishable characteristics from each other motivated by the specific composition of the element and the chemical reactions. In addition, the limited concentrated observation does not take into account the variety of the formation process and behavior of the element. All this makes it reflective whether or not there is life or "abnormal" phenomena.
References
- Kasting JF, Whitmire DP, Reynolds RT. Habitable zones around main sequence stars. Icarus. 1993;101(1):108-128.
[Crossref] [Google Scholar] [PubMed]
- Des Marais DJ, Nuth III JA, Allamandola LJ, et al. The NASA astrobiology roadmap. Astrobiology. 2008;8(4):715-730.
[Crossref] [Google Scholar] [PubMed]
- Artime O, de Domenico M. From the origin of life to pandemics: Emergent phenomena in complex systems. Philos Trans A Math Phys Eng Sci. 2022;380(2227):20200410.
[Crossref] [Google Scholar] [PubMed]
- Longo G, Montévil M. Perspectives on organisms. Berlin: Springer; 2014.
- Walsh DM. Organisms as natural purposes: The contemporary evolutionary perspective. Stud Hist Philos Biol Biomed Sci. 2006;37(4):771-791.
[Crossref] [Google Scholar] [PubMed]
- Braidotti R. Animals, anomalies, and inorganic others. Pmla. 2009;124(2):526-532.