Research
, Volume: 15( 2) DOI: 10.21767/0974-7532.1000154Clinical and Chest Imaging Features of Patients with Human Infection with the Novel Coronavirus 2019
- *Correspondence:
- Xiao-Song Li
Hunan Provincial People’s Hospital/The First Affiliated Hospital of Hunan Normal University
Changsha
China
Tel: +86-731-8392900
E-Mail: lsx0308@sina.com
Received: March 25, 2020; Accepted: June 1, 2020; Published: June 8, 2020
Citation: Fang ZY, Wang LL, Hong XQ, et al. Clinical and Chest Imaging Features of Patients with Human Infection with the Novel Coronavirus 2019. Res Rev Biosci. 2020;15(2):154.
Abstract
Aims: To explore and study the clinical and chest imaging manifestations of patients with the novel coronavirus 2019.
Methods: Using nucleic acid kit to detect the novel coronavirus 2019 virus nucleic acid in patients' respiratory tract samples. In addition, chest CT was used to examine the patient's chest image.
Results: The nucleic acids of the novel coronavirus 2019 were all positive in 5 patients. At the same time, 5 patients were admitted to the hospital and found two lungs involved on CT, which showed ground glass shadow and/or consolidation shadow. Most of them were lower lungs, involving multiple lung segments and lobes. In the consolidation area, air filled bronchogram could be seen.
Conclusion: Patients with pneumonia of the novel coronavirus 2019 have specific chest imaging findings.
Keywords
The novel coronavirus 2019; Pneumonia; Chest CT; Imaging features
Abbreviations:
CT: Computed Tomography; ARDS: Acute Respiratory Distress Syndrome; MODS: Multiple Organ Dysfunction Syndrome; COVID-19: Coronavirus Disease 2019; PSI: Pneumonia Severity Index; CURB-65: Confusion, Urea, Respiratory Rate, Blood Pressure-65
Introduction
Corona Virus Disease 2019 (The novel coronavirus 2019) refers to pneumonia caused by 2019 new coronavirus infection. The new coronavirus is easy to invade the lung tissue of human body and the disease progresses rapidly. Severe pneumonia often complicates with Acute Respiratory Distress Syndrome (ARDS), Multiple Organ Dysfunction Syndrome (MODS), and even death [1,2].
Early diagnosis, early treatment and strengthening the therapy of severe cases are the key to improve the cure rate and reduce the mortality. Imaging examination plays an important role in the evaluation and treatment of severe novel coronavirus pneumonia. In our article 5 cases of Coronavirus Disease 2019 (COVID-19) pneumonia were analyzed and clinical data were summarized.
Data and Methods
Source of data
From January 2020 to February 2020, 5 cases of the novel coronavirus 2019 pneumonia with human infection were collected. The novel coronavirus pneumonia diagnostic and therapeutic plan (Fourth Edition) of The National Health and Family Planning Commission of People's Republic of China was in accordance with the diagnostic criteria [1]. All the respiratory specimens were positive for COVID-19. The suspected cases were excluded.
Clinical data
Among the 5 cases of people infected with the novel coronavirus 2019 pneumonia, there were 1 male and 4 female, aged 45.5-60.5 years, with a median age of 48.0 years; 2 patients had underlying conditions or diseases, including hypertension and schizophrenia, which were not related to this disease; 2 patients had a clear contact history. In retrospective analysis of the clinical data, parameters associated were symptoms, signs, Confusion, Urea, Respiratory Rate, Blood Pressure-65 (CURB-65) score and Pneumonia Severity Index (PSI) score, laboratory examination, imaging features, complications and so on.
Inspection method
Chest CT was performed with Philips britiance 16 slice spiral CT scan. Scanning parameters: tube voltage 120 kv, automatic tube current, pitch 0.9, matrix 512 × 512, layer spacing 5.0 mm. The scanning range was from the apex of lung to bilateral costophrenic angles.
Results
Clinical features
In this group, 5 cases of patients with the novel coronavirus 2019 pneumonia, the first symptoms were mainly fever, cough, fatigue, followed by expectoration and shortness of breath; only one patient had blood and muscle ache in sputum. All of the 5 patients showed low risk in terms of CURB-65 and PSI, decreased in oxygenation index and peripheral blood lymphocyte count, and increase in C-reactive protein, procalcitonin, lactate dehydrogenase, creatine kinase and myoglobin. All patients had severe complications, such as ARDS, followed by septic shock, abnormal liver function, thrombocytopenia and pneumothorax.
Imaging features
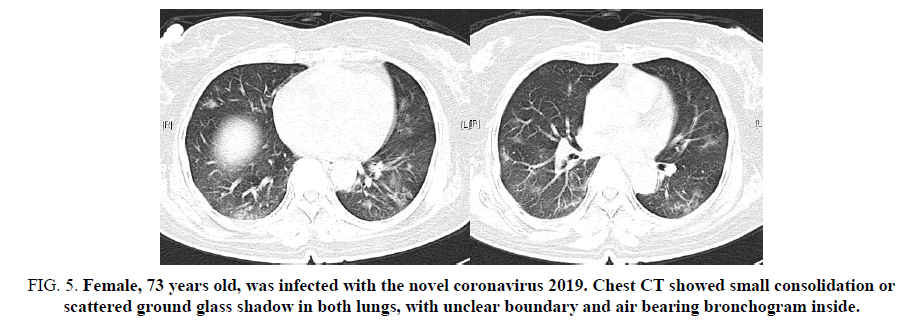
In 5 patients, two lungs were involved on the chest CT, which showed ground glass shadow and (or) consolidation shadow. Most of them were lower lungs, involving multiple lung segments and lobes. In the consolidation area, air filled shadow of bronchus could be seen. In 4 patients, the consolidation of lung gradually increased, and the consolidation of adjacent lung segments fused and spread to adjacent lung lobes (FIG. 1-4); in 1 patient, the two lungs showed small focal consolidation or scattered ground glass shadow (FIG. 5). Mediastinum and hilar lymph nodes were not enlarged.
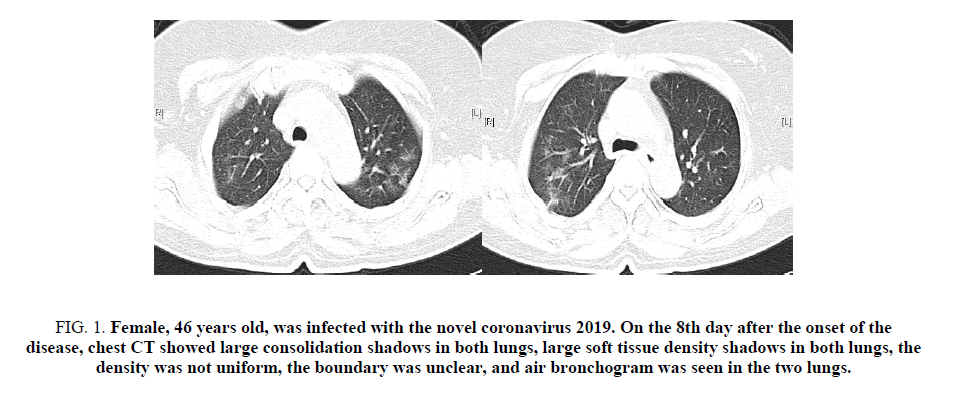
Figure 1. Female, 46 years old, was infected with the novel coronavirus 2019. On the 8th day after the onset of the disease, chest CT showed large consolidation shadows in both lungs, large soft tissue density shadows in both lungs, the density was not uniform, the boundary was unclear, and air bronchogram was seen in the two lungs.
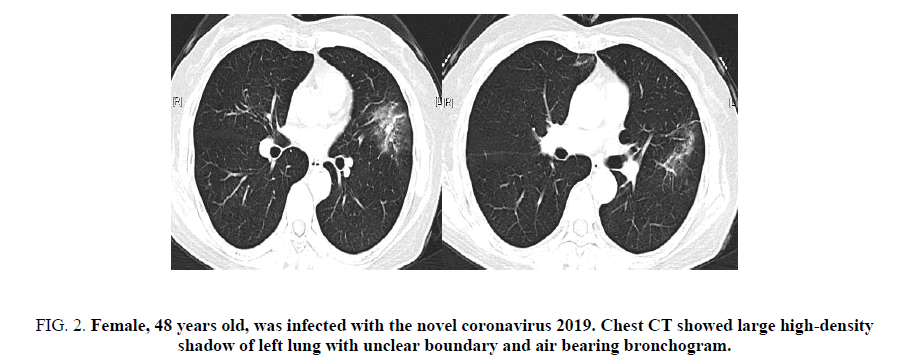
Figure 2. Female, 48 years old, was infected with the novel coronavirus 2019. Chest CT showed large high-density shadow of left lung with unclear boundary and air bearing bronchogram.
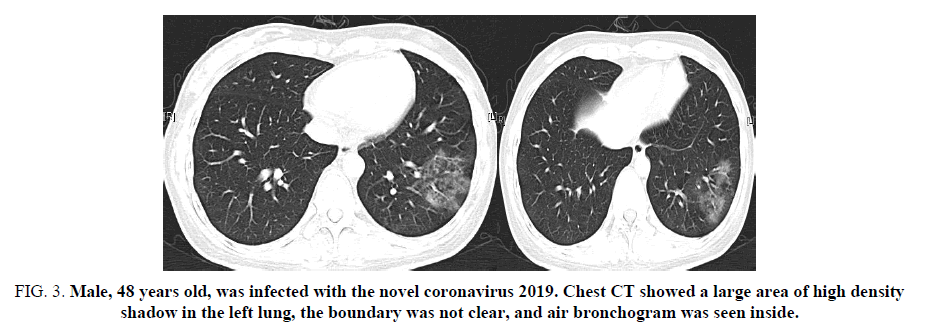
Figure 3. Male, 48 years old, was infected with the novel coronavirus 2019. Chest CT showed a large area of high density shadow in the left lung, the boundary was not clear, and air bronchogram was seen inside.
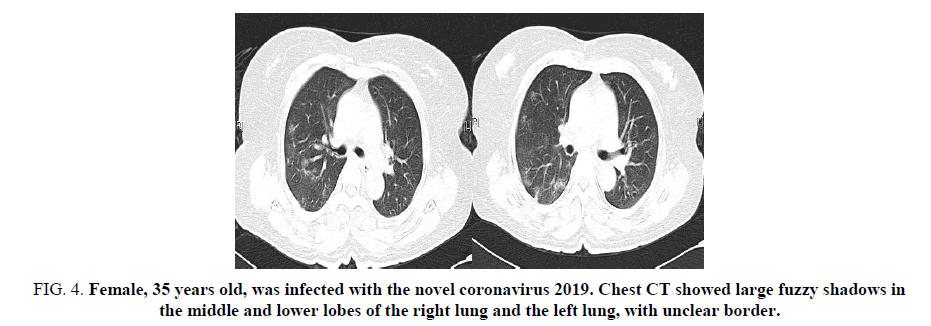
Figure 4. Female, 35 years old, was infected with the novel coronavirus 2019. Chest CT showed large fuzzy shadows in the middle and lower lobes of the right lung and the left lung, with unclear border.
Figure 5. Female, 73 years old, was infected with the novel coronavirus 2019. Chest CT showed small consolidation or scattered ground glass shadow in both lungs, with unclear boundary and air bearing bronchogram inside.
Discussion
The main clinical manifestation of the novel coronavirus 2019 is pneumonia. The disease can evolve rapidly, and many patients develop severe pneumonia in 3-7 days [3,4]. The main symptoms of patients with the novel coronavirus 2019 are influenza like symptoms, most of which are fever, cough, shortness of breath, muscle ache. Some patients with chronic cardiovascular disease have chest distress and palpitation [5,6]. Among the five patients, the average time from onset to hospitalization was 4-6 days, and all of them presented with severe pneumonia. The first symptoms were mainly fever, cough, fatigue, followed by expectoration and shortness of breath.
In this group, 5 patients had low risk of both the CURB-65 (which stands for Confusion, Urea, Respiratory Rate, Blood Pressure and 65 years of age or older) score and the Pneumonia Severity Index (PSI) score. Cossarizza et al. analyzed many patients and found that the actual mortality rate of the low-risk group with CURB-65 score and PSI score was 14.5% and 17.7%, respectively, which was far higher than the theoretical prediction of mortality rate of 1.5% and 1% [1]. Kinross et al. reviewed and analyzed 100 patients with the novel coronavirus 2019. They also found that the actual mortality rate of patients in the low-risk group with CURB-65 score and PSI score was 22.1% and 15.2% respectively, which were far higher than the predicted mortality rate [7]. It can be seen that CURB-65 score and PSI score cannot effectively evaluate the severity of patients with the novel coronavirus 2019 [8].
The patients with the novel coronavirus 2019 were based on the changes of pulmonary interstitial and diffuse alveolar injury. The typical manifestations of chest CT were "consolidation" and "ground glass like changes", which were exudative foci with multiple lung fields. The ground glass like high-density fuzzy shadows with flake solid shadows were mainly distributed in the middle and outer zones of the middle and lower fields of the two lungs. Pulmonary parenchyma and pulmonary stroma are involved at the same time, and pleural effusion may occur later. Severe patients with poor immune function, rapid progress of the disease, prone to ARDS, later can be combined with secondary bacterial and fungal infection [9,10].
In this group, 5 patients were treated with antiviral therapy within 48 hours after admission. 6 days later, all of them were complicated with ARDS. Other complications included septic shock, abnormal liver function and thrombocytopenia, which was consistent with the median time of 14 days in dhama and other studies [11,12].
Conclusion
To sum up, the patients infected with the novel coronavirus 2019 developed rapidly and died of ARDS and MODS in a short time. Imaging plays an important role in the clinical diagnosis and differential diagnosis of human infection with the novel coronavirus 2019. It is necessary to summarize and analyze the epidemiology and clinical characteristics of the confirmed cases, improve the understanding of the disease, and provide clinical experience for the diagnosis and treatment of severe patients with the novel coronavirus 2019 in the future.
Acknowledgments
Zhi-Yong Fang and Li-Le Wang contributed equally to this work.
Conflict of Interest
Authors declared no conflict of interest.
References
- Cossarizza A, De Biasi S, Guaraldi G, et al. SARS?COV?2, the virus that causes COVID?19: cytometry and the new challenge for global health. Cytometry A. 2020;97:340-3.
- Zhu GD, Cao J. Challenges and countermeasures on Chinese malaria elimination programme during the Coronavirus Disease 2019 (COVID-19) outbreak. Chinese J Schistosomias Control. 2020;32:7-9.
- Rosenbaum L. Facing the novel coronavirus 2019 in Italy-ethics, logistics, and therapeutics on the epidemic's front line. N Engl J Med. 2020;382:1873-5.
- Salzberger B, Glück T, Ehrenstein B. Successful containment of COVID-19: the WHO-report on the COVID-19 outbreak in China. Infection. 2020;48:151-3.
- Baden LR, Rubin EJ. COVID-19-the search for effective therapy. N Engl J Med. 2020;382:1851-2.
- Tárnok A. The cholera epidemics in hamburg and what to learn for COVID-19 (SARS-CoV-2). Cytometry A. 2020;97:337-9.
- Kinross P, Suetens C, Gomes DJ, et al. Rapidly increasing cumulative incidence of Coronavirus Disease (COVID-19) in the European Union/European Economic Area and the United Kingdom, 1 January to 15 March 2020. Euro Surveill. 2020;25:2000285.
- Gostin LO, Hodge JG, Wiley LF. Presidential powers and response to COVID-19. JAMA. 2020;323:1547-8.
- Liu D, Li L, Wu X, et al. Pregnancy and perinatal outcomes of women with Coronavirus Disease (COVID-19) pneumonia: a preliminary analysis. AJR Am J Roentgenol. 2020:1-6.
- Thomson G. COVID-19: social distancing, ACE 2 receptors, protease inhibitors and beyond. Int J Clin Pract. 2020:e13503.
- Tolksdorf K, Buda S, Schuler E, et al. Influenza-associated pneumonia as reference to assess seriousness of Coronavirus Disease (COVID-19). Euro Surveill. 2020;25:2000258.
- Dhama K, Sharun K, Tiwari R, et al. COVID-19, an emerging coronavirus infection: advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics. Hum Vaccin Immunother. 2020:1-7.