Research
, Volume: 16( 1) DOI: 10.37532/2277-288X.2026.16(1).205Assessment of Knowledge and Awareness in Patients Consuming Anti-Gout Drugs in the Region of Roorkee
2Department of Biotechnology, Lovely Professional University, Phagwara, Punjab, India
- *Correspondence:
- Muzaffar AliDepartment of Pharmacology, GRD Institute of Management and Technology, Dehradun, Uttarakhand, India E-mail: aliali30924@gmail.com
Received: September 05, 2024, Manuscript No. TSACPI-24-147420; Editor Assigned: September 09, 2024, PreQC No. TSACPI-24-147420 (PQ); Reviewed: September 23, 2024, QC No. TSACPI-24-147420; Revised: February 13, 2026, Manuscript No. TSACPI-24-147420 (R); Published: February 20, 2026, DOI: 10.37532/2277-288X.2026.16(1).205
Citation:Ali M, Ghildiyal M, Tangri P, et al. Assessment of Knowledge and Awareness in Patients Consuming Anti-Gout Drugs in the Region of Roorkee. Acta Chim Pharm Indica. 2026;16(1):205
Abstract
A typical form of inflammatory arthritis that is treated with anti anti-gout drugs is gout. To maximize therapeutic results and increase patient compliance, it is essential to comprehend patients' awareness and knowledge of these medications. With a particular focus on the Roorkee region, this review paper assesses the body of research on patient knowledge and awareness of anti anti-gout medications. Our goals are to draw attention to areas of unmet need, evaluate how education affects patient outcomes, and offer rec ommendations for improving patient comprehension. Genetic predisposition is playing a more and bigger influence. Acute gouty arthritis, intercritical phase, chronic tophaceous gout, and asymptomatic hyperuricemia compri se the clinical picture of gout. Ra diological and laboratory features are used to make the diagnosis. Diagnosis gold standard: identifying distinctive MSU crystals in the synovial polarized light microscopy on a fluid medium. Conventional radiography, ultrasound, dual dual-energy computed tomogr aphy, conventional CT, magnetic resonance imaging, nuclear scintigraphy, and positron emission tomography are examples of imaging modalities. The use of dualdual-energy CT and ultrasonography has advanced significantly, which will undoubtedly have an impact on clinical research as well as di agnosis, staging, and follow follow-up. Gout management encompasses controlling flare flare-ups, managing chronic gout and preventing flare flare-ups, and managing comorbidities. Newer medications are proving to be effective and serving as a s upplement to older ones in the pharmacological arsenal. A change in food and lifestyle, patient education, and stopping hyperuricemia medications are all crucial aspects of its management.
Keywords
NSAIDs; Colchicine; Allopurinol; Gout
Introduction
The symptoms of gout include high blood sugar levels and urate crystal buildup in joints, which causes sudden inflammation. Medicinal treatments for gout often include colchicine, Non-Steroidal Anti-Inflammatory Medicines (NSAIDs), and urate-lowering medications such febuxostat and allopurinol. Even though there are effective treatments available, patients frequently lack awareness and information about these medications, which can have an impact on treatment success and adherence [1,2]. This review aims to identify shortcomings and suggest changes by evaluating the knowledge and awareness of patients in Roorkee who are taking anti-gout medications.
MSU crystals can create tophi in many tissues, but they are most commonly found in and surrounding joints. The primary method for diagnosing gout is to identify the pathognomonic MSU crystals through joint fluid aspiration or tophi aspirate. Gout's early manifestation is an intense joint irritation that is promptly reduced by colchicine or NSAIDs. Tophi and renal stones are late appearances. Reducing SUA.
The major objective in treating gout is to get serum uric acid levels below the deposition threshold, either by dietary changes or medication. As a result, MSU crystals dissolve and stop additional attacks [3,4].
Gout this metabolic condition is typified by hyperuricemia, with a normal plasma urate of 2-4 mg/dl. The byproduct of purine metabolism, uric acid, is poorly soluble in water, particularly at low pH levels. High blood levels cause tophi, or precipitation and depositing in the kidney, joints, and subcutaneous tissue. Secondary hyperuricemia can arise from increased metabolism of nucleic acids and uric acid synthesis in leukemias, lymphomas, and polycythemia, particularly in cases treated with chemotherapy or radiation therapy [5].
Drug-induced: Levodopa, thiazides, furosemide, pyrazinamide, and ethambutol decrease the kidney's excretion of uric acid.
Treatments for gout include: For severe gout NSAIDs colchicine corticosteroids for hyperuricemia/chronic gout uricosurics inhibitors of synthesis probenecid theopurinol sulfinpyrazone febuxostat.
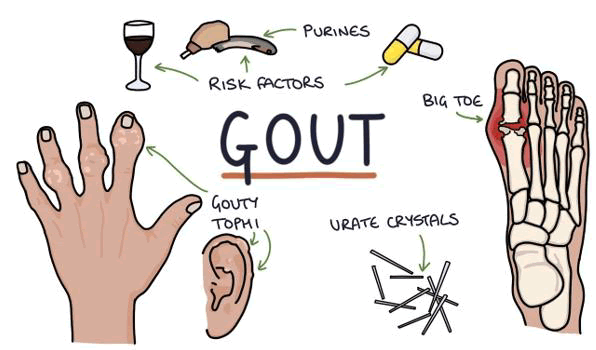
The most common symptom of acute gout is the abrupt development of severe inflammation in a small joint (the metatarso-phalangeal joint of the great toe) as a result of urate crystals precipitating in the joint space. The joint becomes very painful, swollen, and red; it needs to be treated very away (Figure 1).
FIG. 1. Gout symptoms.
Materials and Methods
A thorough literature search was carried out using databases including PubMed, Google Scholar, and Scopus in order to produce this study. The research conducted in Roorkee or similar places was given particular importance in the search, which concentrated on studies concerning patient knowledge and awareness of anti-gout drugs [6,7]. Relevance, methodological rigor, and contribution to the understanding of patient knowledge were the determining factors in the selection of studies.
Patient knowledge and awareness: General insights
Understanding of anti-gout medications
NSAIDs: Although most patients know that NSAIDs reduce pain and inflammation, they might not be aware of all the possible interactions and side effects.
Colchicine: Lack of knowledge regarding colchicine's mechanism of action and adverse effects frequently results in overuse and improper dosage [8].
Xanthine oxidase inhibitors: The synthesis of uric acid is facilitated by the enzyme xanthine oxidase, which is inhibited by medications such as febuxostat and allopurinol.
Urate-lowering agent: Febuxostat and allopurinol are necessary for the long-term treatment of gout. But often, patients don't know enough about their part in lowering uric acid levels, adherence tactics, and possible side effects [9].
Interleukin-1 inhibitors: When other treatments are ineffective for severe cases of gout, physicians may prescribe biologics like canakinumab or anakinra. These medications focus on particular inflammatory pathways.
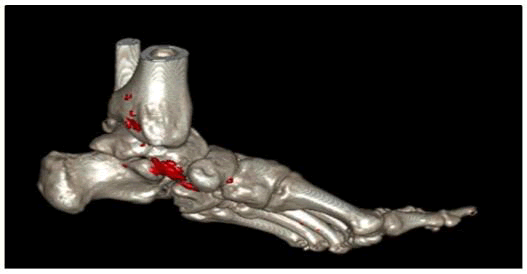
It's crucial to remember that lifestyle adjustments, like as eating differently and keeping a healthy weight, are frequently advised in addition to prescription drugs to treat gout. It may also be helpful to stay hydrated and steer clear of meals high in purines, which can cause the body to produce more uric acid [10,11]. For an accurate diagnosis and the best course of action for your particular circumstances, always seek the advice of a healthcare professional (Figure 2).
FIG. 2. 3D CT reconstruction of a human foot showing ankle joint pathology.
Mechanism of action
Xanthine oxidase inhibitors
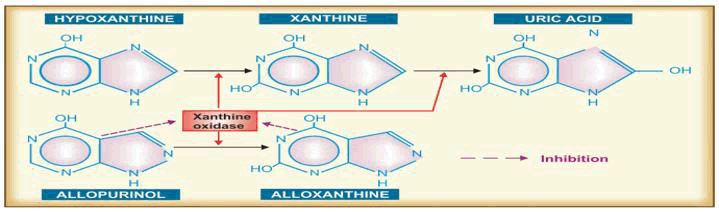
Drugs known as xanthine oxidase inhibitors function by preventing the xanthine oxidase enzyme from doing its job [12]. Because of the important role this enzyme plays in the conversion of purines into uric acid, uric acid production can be decreased by blocking its activity. One of the most important tactics in the treatment of diseases like gout is lowering uric acid levels.
The following is a condensed description of how xanthine oxidase inhibitors work:
Normal purine metabolism: Purines are organic compounds that are present in the body and in some foods. They are reduced to a substance known as xanthine, which the enzyme xanthine oxidase then transforms into uric acid.
Xanthine oxidase inhibition: Allopurinol and febuxostat are examples of xanthine oxidase inhibitors that interfere with this process by preventing xanthine oxidase from doing its job [13,14]. One of xanthine oxidase's typical substrates, hypoxanthine, has a structural homolog in allopurinol.
Allopurinol: The body transforms allopurinol into oxypurinol, a strong xanthine oxidase inhibitor. It lessens the conversion of these substrates into uric acid by competing with hypoxanthine and xanthine for the active site of xanthine oxidase.
Febuxostat: A non-purine selective inhibitor of xanthine oxidase is called fruxostat. Additionally, it functions by inhibiting the activity of the enzyme, which stops purines from being converted to uric acid [15].
Reduced uric acid production: Through the inhibition of xanthine oxidase, these drugs lower uric acid generation. For those suffering from disorders like gout, lowering uric acid levels is advantageous since it lessens the risk of urate crystals forming in the joints, which can result in discomfort and inflammation.
Unfortunately, I am unable to supply images. However, textbooks on biochemistry or pharmacology, scholarly publications, or internet learning materials can provide diagrams and drawings of the purine metabolic route and the mechanism of action of xanthine oxidase inhibitors (Figure 3).
FIG. 3. Uric acid synthesis and the action of allopurinol.
Compliance and adherence issues
Research shows that a lack of knowledge about the chronic nature of gout and the necessity of continuous treatment is associated with poor adherence to anti-gout drugs [16-18]. When their symptoms subside, patients may quit taking their medications, not understanding how crucial it is to keep their uric acid levels within the desired range.
Regional focus: Roorkee
Demographic and socioeconomic context
The population of Roorkee, an Indian city in Uttarakhand, is diverse and has differing access to healthcare and education. When it comes to medication adherence and patient awareness, socioeconomic considerations are important. Inadequate healthcare resources and lower literacy rates can also lead to knowledge gaps [19,20].
Local studies and findings
Local studies have highlighted several issues
Knowledge gaps: Many Roorkee patients are unaware of the chronic nature of gout and the necessity of ongoing therapy.
Healthcare access: Patients' access to follow-up and education is impacted by limited access to specialist healthcare services.
Cultural factors: Patients' opinions and adherence to traditional anti-gout medicines may be influenced by traditional medicine practices and beliefs.
Impact of educational interventions
It has been demonstrated that educational initiatives increase patient understanding and gout treatment adherence. Better medication adherence can be encouraged by interventions including community health workshops, informational leaflets, and patient counseling that help close the knowledge gap.
Results
Section I: Description of the demographic characteristics of gout patients (Table 1 and Figure 4).
| S. no | Demographic variables | Frequency (f) | Percentage (%) |
| 1 | Age in years | ||
| 30 ± 40 | 5 | 16 | |
| 41 ± 50 | 12 | 40 | |
| 51 ± 60 | 10 | 33 | |
| 61 and above | 6 | 20 | |
| 2 | Sex | ||
| Male | 20 | 66 | |
| Female | 10 | 33 | |
| 3 | Types of family | ||
| Joint family | 17 | 57 | |
| Nuclear family | 13 | 43 | |
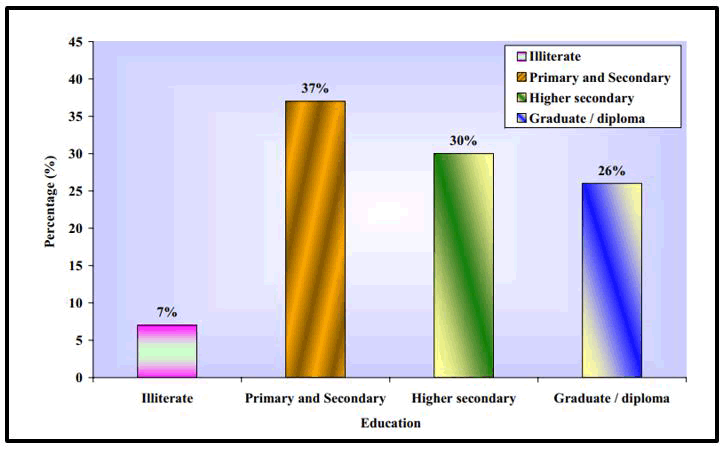
| 4 | Education | ||
| Illiterate | 2 | 7 | |
| Primary and Secondary | 11 | 37 | |
| Higher secondary | 9 | 30 | |
| Graduate/Diploma | 8 | 26 | |
| 5 | Occupation | ||
| Agriculture | 4 | 13 | |
| Business | 7 | 23 | |
| Self-employment | 5 | 17 | |
| Teacher | 5 | 17 | |
| Others | 9 | 30 | |
| 6 | Income | ||
| 2000 ± 4000 | 13 | 43 | |
| 4000 ± 6000 | 14 | 47 | |
| 6001 ± 8000 | 2 | 7 | |
| Above 8000 | 1 | 3 | |
| 7 | Source of information | ||
| Family members | 3 | 10 | |
| Neighbor | 19 | 63 | |
| Telecommunication | 8 | 27 | |
| Health education | 0 | 0 | |
| 8 | Types of food habits | ||
| Vegetarian | 5 | 16 | |
| Non-vegetarian | 25 | 83 | |
| 9 | Types of gout | ||
| Acute gout | 12 | 40 | |
| Mild | 18 | 60 | |
| Moderate | |||
TABLE. 1 Description of demographic variables of patients with gout (n=30).
FIG. 4. Distribution of demographic variables according to the education status of the patients.
Discussion
Enhancing patient education
Pharmacist involvement: By giving patients comprehensive information regarding drug use, side effects, and the value of adherence, pharmacists can play a critical role in patient education.
Healthcare workshops: Putting together community workshops to increase knowledge about gout treatment and the function of gout medications.
Educational materials: To increase patient comprehension, easily understood materials should be developed in the local language. Improving healthcare access The Roorkee healthcare system needs to be improved, and this includes making instructional materials and specialized gout management services available.
Conclusion
There are notable gaps in the knowledge and understanding of anti-gout medication users in Roorkee, which affects how well their treatments work. By addressing these gaps with focused educational interventions and expanding access to healthcare, it will be possible to increase patient comprehension and adherence to anti-gout medications, which will eventually result in better gout management and higher quality of life for patients.
References
- Emmerson BT. The management of gout. N Engl J Med. 1996;334(7):445-451.
[Crossref] [Google Scholar] [PubMed]
- Pascual E, Sivera F. Time required for disappearance of urate crystals from synovial fluid after successful hypouricaemic treatment relates to the duration of gout. Ann Rheum Dis. 2007;66(8):1056-1058.
[Crossref] [Google Scholar] [PubMed]
- Singh JA. Challenges faced by patients in gout treatment: A qualitative study. J Clin Rheumatol. 2014;20(3):172-174.
[Crossref] [Google Scholar] [PubMed]
- Kuo CF, Grainge MJ, Zhang W, et al. Global epidemiology of gout: Prevalence, incidence and risk factors. Nat Rev Rheumatol. 2015;11(11):649-662.
[Crossref] [Google Scholar] [PubMed]
- Mccarty DJ, Hollander JL. Identification of urate crystals in gouty synovial fluid. Ann Intern Med. 1961;54(3):452-460.
[Crossref] [Google Scholar] [PubMed]
- Mandal AK, Mount DB. The molecular physiology of uric acid homeostasis. Annu Rev Physiol. 2015;77(1):323-345.
[Crossref] [Google Scholar] [PubMed]
- Kamei K, Konta T, Hirayama A, et al. A slight increase within the normal range of serum uric acid and the decline in renal function: Associations in a community-based population. Nephrol Dial Transplant. 2014;29(12):2286-2292.
[Crossref] [Google Scholar] [PubMed]
- Torres RJ, Puig JG. Hypoxanthine-guanine phosophoribosyltransferase (HPRT) deficiency: Lesch-Nyhan syndrome. Orphanet J Rare Dis. 2007;2:1-10.
[Crossref] [Google Scholar] [PubMed]
- Reginato AM, Olsen BR. Genetics and experimental models of crystal-induced arthritis. Lessons learned from mice and men: Is it crystal clear?. Curr Opin Rheumatol. 2007;19(2):134-145.
[Crossref] [Google Scholar] [PubMed]
- Kanbara A, Seyama I. Effect of urine pH on uric acid excretion by manipulating food materials. Nucleosides Nucleotides Nucleic Acids. 2011;30(12):1066-1071.
[Crossref] [Google Scholar] [PubMed]
- Towiwat P, Li ZG. The association of vitamin C, alcohol, coffee, tea, milk and yogurt with uric acid and gout. Int J Rheum Dis. 2015;18(5):495-501.
[Crossref] [Google Scholar] [PubMed]
- Mahmoud HH, Leverger G, Patte C, et al. Advances in the management of malignancy-associated hyperuricaemia. Br J Cancer. 1998;77(4):18-20.
[Crossref] [Google Scholar] [PubMed]
- Emmerson B. Hyperlipidaemia in hyperuricaemia and gout. Ann Rheum Dis. 1998;57(9):509-510.
[Crossref] [Google Scholar] [PubMed]
- Dessein PH, Shipton EA, Stanwix AE, et al. Beneficial effects of weight loss associated with moderate calorie/carbohydrate restriction, and increased proportional intake of protein and unsaturated fat on serum urate and lipoprotein levels in gout: A pilot study. Ann Rheum Dis. 2000;59(7):539-543.
[Crossref] [Google Scholar] [PubMed]
- Ichida K, Matsuo H, Takada T, et al. Decreased extra-renal urate excretion is a common cause of hyperuricemia. Nat Commun. 2012;3(1):764.
[Crossref] [Google Scholar] [PubMed]
- Enomoto A, Endou H. Roles of organic anion transporters (OATs) and a urate transporter (URAT1) in the pathophysiology of human disease. Clin Exp Nephrol. 2005;9:195-205.
[Crossref] [Google Scholar] [PubMed]
- Bobulescu IA, Moe OW. Renal transport of uric acid: Evolving concepts and uncertainties. Adv Chronic Kidney Dis. 2012;19(6):358-371.
[Crossref] [Google Scholar] [PubMed]
- Han J, Liu Y, Rao F, et al. Common genetic variants of the human uromodulin gene regulate transcription and predict plasma uric acid levels. Kidney Int. 2013;83(4):733-740.
[Crossref] [Google Scholar] [PubMed]
- Cho SK, Kim S, Chung JY, et al. sDiscovery of URAT1 SNPs and association between serum uric acid levels and URAT1. BMJ Open. 2015;5(11):009360.
[Crossref] [Google Scholar] [PubMed]
- Tan PK, Ostertag TM, Miner JN. Mechanism of high affinity inhibition of the human urate transporter URAT1. Sci Rep. 2016;6(1):34995.
[Crossref] [Google Scholar] [PubMed]