Original Article
, Volume: 11( 4)Supramolecular Complexes of Glycyrrhizic Acid, its Monoammonium Salt with Diterpenoid Lagochilin and their Hemostatic Activity
- *Correspondence:
- Matchanov AD Sadykov Institute of Bioorganic Chemistry, Academy of Sciences of Uzbekistan 100125, Mirzo Ulughbek str. 83, Tashkent, Uzbekistan
Tel: 998711623540; E-mail: olim_0172@mail.ru
Received Date: September 1, 2017 Accepted Date: September 4, 2017 Published Date: September 8, 2017
Citation: Matchanov AD, Zaynutdinov UN, Islamov AKH, et al. Supramolecular Complexes of Glycyrrhizic Acid, its Monoammonium Salt with Diterpenoid Lagochilin and their Hemostatic Activity. Biochem Ind J. 2017;11(4):118
Abstract
Data on the synthesis of supramolecular complexes of lagochilin with glycyrrhizic acid and its monoammonium salt, and their haemostatic activity are presented. It is shown that the hemostatic activity of complexes depends not only on the nature of the ligand and guest molecule, but also on their relationship.
Keywords
Biochemical parameters; Donkeys; West Africa; Working animals
Introduction
The donkey was, in the past, a faithful companion of commercial expeditions of peddlers in West Africa. Before to the introduction of the automobile, the donkey was used in the transport of salt, cola, cotton and many other commodities between coastal regions and landlocked areas such as Mali, Burkina Faso and Niger [1].
Nowadays, donkeys still play an important socio-economic role both in the rural and urban areas. In the rural area, the donkeys are used for crop production in the farm, transport of goods and people. In the cities, the carts with asine traction are used for garbage collection and the transport of building materials. Donkeys are considered as working animal for any task owing to their natural rusticity and endurance.
Donkeys, like other working animals, are used because of their muscular strength and endurance. The search for performance exposes them to organic lesions and often to the detriment of their well-being. Intense muscular effort can cause muscle damage that can lead to increased enzymes activities such as Aspartate aminotransferase (AST) and creatine kinase (CK) [2,3]. Fatigue, maltreatment, lack of veterinary care and malnutrition in working animals such as donkeys can cause changes in biochemical parameters [4].
Despite their important socio-economic role, very few studies have been devoted to the establishment of the reference values of the clinical parameters of working animals in West Africa, especially in donkeys with a large sample extended to various countries. Hence, the overall objective of the study aimed at establishing the biological and clinical constants in West African donkeys breed.
Material and Methods
Study area
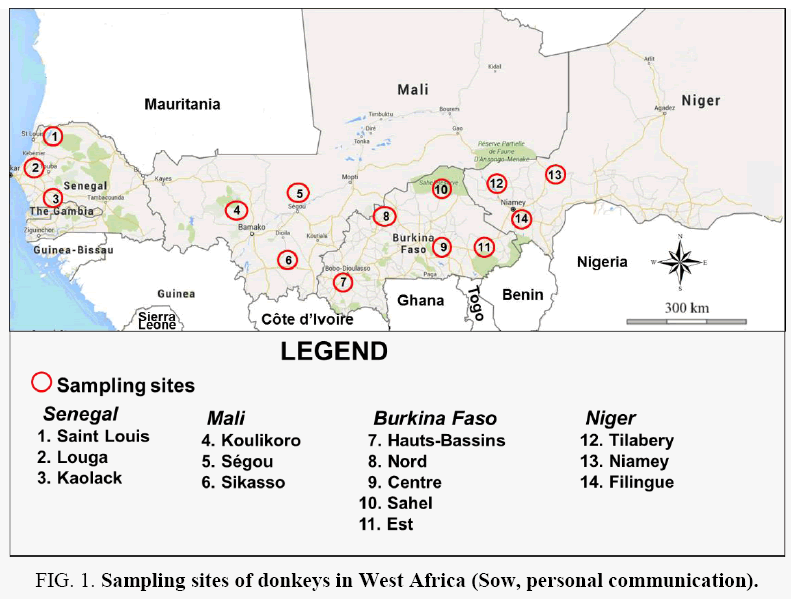
This study was carried out in 4 West African countries: Burkina Faso, Mali, Niger and Senegal. For Mali, Niger and Senegal, sampling was done in three well-defined sites in each country, drawn at random from the exhaustive list of their administrative regions; five sites were selected for Burkina Faso [Figure 1]. All sampling sites are agricultural areas for excellence where donkeys are used as agricultural auxiliaries and are essential for family farms production.
Sampling
A total of 690 donkeys were selected amongst of which 510 males and 180 females distributed in the various localities [Table 1]. The age was estimated based on dentition as described by Muylle et al. [5]. For a good interpretation of the influence of age on biochemical parameters, the donkeys were divided into 3 age groups: group 1 [1-3 years]; group 2 [3-5 years] and group 3 (>5 years).
| Country | N | Nb males | Nb females | Age (years) | Weight (kg) | Mean BCS |
|---|---|---|---|---|---|---|
| Burkina Faso | 150 | 118 | 32 | 6.88±3.6 | 114.47±21.1 | 2.52±0.8 |
| Mali | 90 | 64 | 26 | 7.12±3.4 | 117.82±22.6 | 2.2±0.8 |
| Niger | 150 | 134 | 16 | 7.52±3.2 | 130.77±15.4 | 2.86±0.7 |
| Senegal | 300 | 194 | 106 | 4.72±2.7 | 123.74±22.3 | 2.67±0.6 |
| Total | 690 | 510 | 180 | 6.12±3.4 | 122.52±21.5 | 2.62±0.7 |
| Nb: Number, BCS: Body Condition Score. | ||||||
Table 1: Repartition of the studied population of donkeys in West Africa.
All sampled animals were apparently healthy, subjects less than 1 year old and subjects too old were discarded. Each animal was weighed using an electronic scale (RUDDWEIGH KM-2E Electronic Weighing System®). The nutritional status of the donkeys was assessed using a body condition score grid (BCS) established by Vall et al. [6].
A blood sample was collected from the jugular vein puncture on a dry glass tube. Blood samples were allowed to clot, and serum was separated by centrifugation at 1,500 xg for 10 min. Then, samples were frozen and stored at -20°C until biochemical analyses could be performed.
Biochemical analyses
Biochemical analyses determined levels for metabolites (total proteins, albumin, creatinine, and urea), enzyme (aspartate aminotransferase (AST)), and ions (calcium, magnesium, and phosphorus). Biochemical analyses were carried out using commercial kits (BIOSYSTEMS® SA, Barcelona, Spain), and the experimental protocols were provided by the manufacturers. The colorimetric reactions were measured using spectrophotometer (BIOSYSTEMS® SA, Barcelona, Spain).
Statistical analyses
Data were computed on Excel® and were analyzed using STATA SE 9.2® software. The means and standard deviations were calculated for each of the biochemical parameters. Student's t test and ANOVA were used to compare values of biochemical parameters in the different groups of donkeys. Differences were considered to be statistically significant with values of p<0.05.
Results
Biochemical parameters of apparently healthy donkeys
Biochemical parameters values of apparently healthy West African donkeys breed were comparable to the reference values found in donkeys elsewhere except the values of creatinine and AST [Table 2].
| Biochemical parameters | Mean values | Reference values |
|---|---|---|
| Total Proteins (g/l) | 64.96 ± 10.66 | 58-82a |
| Albumin (g/l) | 26.94 ± 8.90 | 20-34a |
| Creatinine (µmol/l) | 109.58 ± 31.54* | 159.13 ± 12.37b |
| Urea (mmol/l) | 6.56 ± 2.09 | 1.9-7.6a |
| Calcium (mmol/l) | 2.99 ± 0.7 | 2.13 ± 0.04b |
| Phosphorus (mmol/l) | 1.25 ± 0.6 | 0.90 ± 0.12b |
| Magnesium (mmol/l) | 1.44 ± 0.6 | 1.25 ± 0.15c |
| AST (UI/l) | 237.08 ± 95.11* | 295.81 ± 62.79b |
| a: (French et al., 1995), b: (Mori et al., 2003), c: (Al-Busadah et Homeida, 2005), *Values lower than the values of the literature. | ||
Table 2: Mean values of biochemical parameters of apparently healthy West African donkeys.
Variation of biochemical parameters by sex
Mean values of the biochemical parameters of ions (calcium, phosphorus and magnesium) were higher in females than in males (p<0.05). However, the value of AST was significantly lower in females compared to males [Table 3].
| Biochemical parameters | Males (n=510) | Females (n=180) | t-test | p-value |
|---|---|---|---|---|
| Total Proteins (g/l) | 65.53±10.4 | 63.96±11.3 | 1.69 | 0.092 |
| Albumin (g/l) | 26.63±7.4 | 26.61±7.8 | 0.03 | 0.975 |
| Creatinine (µmol/l) | 110.19±32.6 | 107.87±28.4 | 0.85 | 0.802 |
| Urea (mmol/l) | 6.64±2.2 | 6.36±1.9 | 1.56 | 0.94 |
| Calcium (mmol/l) | 2.95±0.6 | 3.11±0.8 | 2.65 | 0.004* |
| Phosphorus (mmol/l) | 1.21±0.6 | 1.37±0.6 | 3.32 | 0.001* |
| Magnesium (mmol/l) | 1.39±0.6 | 1.58±0.8 | 3.54 | 0.0002* |
| AST (UI/l) | 244.98±98.7 | 215.14±80.8 | 3.62 | 0.0002* |
| *p<0.05, significant difference. | ||||
Table 3: Variation of biochemical parameters by sex.
Variation of biochemical parameters by country
Depending on the country, there were significant differences (p<0.05) for all parameters [Table 4]. However, none of these values is pathological. The value of AST was higher among donkeys in Burkina Faso than in other countries.
| Biochemical parameters | Burkina (n=150) | Mali (n=90) | Niger (n=150) | Senegal (n=300) |
|---|---|---|---|---|
| Total Proteins (g/l) | 65.32 ± 6.9a | 67.76 ± 9.5b | 66.34 ± 9.1c | 63.62 ± 12.9d |
| Albumin (g/l) | 21.21 ± 3.3a | 22.03 ± 3.6a | 31.32 ± 6.4c | 28.36 ± 8.0d |
| Creatinine (µmol/l) | 105.47 ± 24.4a | 132.52 ± 29.7b | 110.64 ± 32.2c | 104.24 ± 32.0d |
| Urea (mmol/l) | 6.59 ± 1.5a | 5.85 ± 1.4b | 8.31 ± 2.4c | 5.90 ± 1.8d |
| Calcium (mmol/l) | 2.87 ± 0.4a | 2.66 ± 0.6b | 2.87 ± 0.4c | 3.22 ± 0.8a |
| Phosphorus (mmol/l) | 0.97 ± 0.5a | 1.20 ± 0.8b | 1.25 ± 0.39c | 1.41 ± 0.6d |
| Magnesium (mmol/l) | 1.13 ± 0.3a | 1.42 ± 0.4b | 1.53 ± 0.32c | 1.55 ± 0.9c |
| AST (UI/l) | 294.23 ± 75.6a | 238.42 ± 68.8b | 231.44 ±70.72c | 210.55 ± 107.4d |
| Means followed by same letters in rows did not differ significantly at p<0.05. | ||||
Table 4: Variation of biochemical parameters by country.
Variation of biochemical parameters according to nutritional status
There were significant differences between the values of almost all parameters except phosphorus and magnesium [Table 5]. Mean values of AST, total protein and albumin were significantly higher in subjects with a body condition score (BCS 4).
| Biochemical parameters | BCS 1 (n= 26) | BCS 2 (n= 279) | BCS 3 (n=18) | BCS 4 (n= 67) |
|---|---|---|---|---|
| Total Proteins (g/l) | 63.53 ± 10.3a | 65.41 ± 10.9b | 64.74 ± 10.4c | 66.34 ± 11.5d |
| Albumin (g/l) | 18.97 ± 3.1a | 26.12 ± 7.8b | 27.49 ± 7.3c | 27.56 ± 6.2d |
| Creatinine (µmol/l) | 115.89 ± 27.8a | 108.17 ± 30.7b | 109.03 ± 31.6c | 115.69 ± 35.6d |
| Urea (mmol/l) | 6.05 ± 1.9a | 6.23 ± 2.0b | 6.84 ± 2.2c | 6.89 ± 2.0d |
| Calcium (mmol/l) | 2.68 ± 0.4a | 3.01 ± 0.7b | 3.02 ± 0.6c | 2.95 ± 0.7d |
| Phosphorus (mmol/l) | 0.92 ± 0.7 | 1.19 ± 0.6 | 1.33 ± 0.6 | 1.26 ± 0.4 |
| Magnesium (mmol/l) | 1.19 ± 0.4 | 1.40 ± 0.6 | 1.51 ± 0.7 | 1.34 ± 0.5 |
| AST (UI/l) | 210.77 ± 63.9a | 234.03 ± 98.9b | 235.86 ± 94.4c | 266.80 ± 87.4d |
| Means followed by same letters in rows did not differ significantly at p<0.05. | ||||
Table 5: Variation of biochemical parameters according to nutritional status (BCS).
Variation of biochemical parameters by age group
Significant variations were noted between the mean values of most biochemical parameters according to the age group of donkeys. However, there was no significant variation for phosphorus and magnesium. Although, mean values for creatinine, phosphorus and calcium were higher in the younger donkeys (group 1). In adult subjects over 5 years of age (group 3) mean values of total protein and AST were higher than in younger subjects [Table 6].
| Biochemical parameters | Group 1 (n= 92) | Group 2 (n= 228) | Group 3 (n= 370) |
|---|---|---|---|
| Total Proteins (g/l) | 63.39±12.90a | 64.08±11.08b | 65.89±9.68c |
| Albumin (g/l) | 27.49±11.36a | 27.53±9.50b | 26.45±7.74c |
| Creatinine (µmol/l) | 112.68±34.04a | 107.39±32.64b | 110.16±30.17c |
| Urea (mmol/l) | 5.98±1.86a | 6.56±2.13b | 6.71±2.10c |
| Calcium (mmol/l) | 11.01±5.04a | 10.45±4.39b | 9.66±4.55c |
| Phosphorus (mmol/l) | 4.62±2.71a | 3.72±2.04b | 3.13±1.19c |
| Magnesium (mmol/l) | 3.27±2.18 | 3.36±2.15 | 2.91±1.41 |
| AST (UI/l) | 214.82±113.19a | 233.18± 99.36b | 245.13±86.26c |
| Group 1: [1-3 years]; Group 2: [3-5 years]; Group 3: (>5 years). Means followed by same letters in rows did not differ significantly at p<0.05. | |||
Table 6: Variation of biochemical parameters by age group.
Discussion
In general, all values of the biochemical parameters obtained in donkeys in West Africa were close to those obtained in European, Latin American and Middle Eastern donkeys [7-11]. However, the values of AST and creatinine were lower than the values obtained elsewhere. These two parameters evolve according to muscular activity and muscle mass respectively. The low values in the West African donkeys may be explained by their modest body mass. Indeed, a donkey from West Africa weighs about 122 kg [12] while donkeys of exotic breeds are generally heavier; the donkey of Poitou and the donkey of the Balkans donkey weigh on average 350 kg, 160 kg, respectively [13,14].
Mean values of creatinine and AST of donkeys varied significantly according to the country, BCS and age groups (p<0.05). The variation in creatininemia is due to the difference in weight between donkeys. Moreover, its plasma concentration may vary according to age but also to the intensity of the muscular effort [15,16].
The variation in AST is mainly due to muscular activity. Although donkeys in Burkina Faso and Mali had lower live weights than Niger and Senegal, their AST values were higher than those of the two latter countries. This increase in AST values in these countries could be explained by the fact that donkeys are more exploited in these two countries in the more laborious work (plowing and pulling of asine carts). On the other hand in Senegal, donkeys are used by children and the most important tasks are carried out by horses. Similarly, in Niger, donkeys are used by women where they carry out water chores and transport of people. The AST assay could thus allow gauging the welfare of traction donkeys.
This is corroborated by the fact that the AST values were higher in the donkeys of BCS1 compared to those of BCS2, BCS3 and BCS4. The donkeys of BCS4 (fatty donkeys) had the lowest AST value compared to malnourished donkeys or with low body condition score. Thus the AST value also varied significantly (p<0.05) by age group. Donkeys over 5 years of age had significantly higher values than younger subjects. Donkeys within this age group are used daily and for long hours to pull the asine carts, muscle activity is therefore more intense than the younger subjects. Girardi et al. [17] found that in Pêga donkeys in Brazil, AST values in subjects over 3 years of age were higher than in younger donkeys.
Similarly, total protein values increased significantly (p<0.05) according to age of the donkeys. This is consistent with the results of previous studies [7,18,19]. This variation is due to an increase in the globulin portion of the proteins because the albumin portion decreased with age. The increase in the globulin fraction with age may be due to the improvement of immunocompetence in response to environmental adversity [17].
In contrast to total proteins, calcium and phosphorus concentrations were higher in young than in older subjects. This could be explained by the greater capacity of young subjects to mobilize calcium and phosphorus for their growth [15]. This corroborates the results obtained by Caldin et al. [20] which showed that calcium and phosphorus concentrations were higher in young than in older subjects, but also the results of Orlandi et al. [21] and Jordana et al. [10], who have actually described calcium and phosphorus concentrations decreasing with age of donkeys, this decline with age probably corresponds to a reduction in bone metabolism [22].
The mean value of AST varied significantly between the two sexes (p=0.0002). On the one hand, the males had a higher live weight than the females, and on the other hand the males were more used in the traction for field work. The high value of AST in males may be due to more intense muscle activity. Previous studies have shown that the value of AST in female donkeys was lower than in males [8,18]. However, some studies have proved the opposite with higher AST values in females [17]. However, Sow et al. [23] found that in donkeys in the agropastoral zone of Burkina Faso there was no significant difference in the average values of AST between the two sexes. Indeed, in this region, and indifferently from the sex, both male and female donkeys are used for traction, and therefore there are all subjected to the same intensity of muscular activity.
Meanwhile, average values of magnesium, calcium and phosphorus were significantly higher in females than males (p<0.05). Our results were different from those of Girardi et al. [17] who found that the values of these ions were lower in females than in males. However, there are controversial results regarding the variation in ions concentration regarding the sex of donkeys. Indeed, Homeida [8] found no significant difference in magnesium concentration between both sexes, but these authors noted that calcium concentrations were higher in males than in females.
All biochemical parameters were significantly different in donkeys from one country to another. This variation may be due to different climatic conditions between countries. It may also be related to the sampling period. Indeed, if during the rainy season the samples were collected, the animals are subjected to intense farm work and have a very sufficient diet with all the necessary nutrients. While if sampling is done during dry season, the donkeys are still over exploited and moreover they are malnourished because of the natural forage become rare and nutrient-poor. Therefore, we stated that the variation in biochemical parameters is due to the muscular activity performed by the donkeys and the quality of the forage.
Conclusion
The results of this study showed that in West African donkeys the age, nutritional status (BCS), locality and sex of the animal influenced some reference values of biochemical parameters. Therefore, it is essential to establish reference ranges of values specific to local donkeys breed in West Africa. The differences between the results of the local donkeys and those from elsewhere are due to zootechnical and environmental factors. The setting of the biochemical parameters range of values will serve as reference values for clinicians for a proper interpretation of biochemical analyses for better healthcare of donkeys.
Conflict of Interest
The authors declare that there is no conflict of interest.
Authors Contributions
AS, AB and MK drafted the experimental protocol and carried out the statistical analyzes and interpretation of the results, CRR, GP and MMD carried out the sampling and laboratory analyzes. All authors contributed to the writing of the manuscript.
Acknowledgement
We express our gratitude to the Director General of EISMV for providing good working conditions. We thank the veterinary services of Burkina Faso and Senegal, the Faculty of Agronomy of the Abdou Moumouni University of Niger and the SPANA in Mali and all donkey owners for their collaboration on during field surveys.
References
- Harre D. Businessmen in Côte d'Ivoire: The integration of the Malinkés of Kabadougou into the contemporary economy. In: Great traders in West Africa: Logic and practices of a group of contemporary businessmen. Grégoire E. and Labazée P. (eds.), Paris, Karthala. 1999.
- Kaneko JJ, Harvey JW, Bruss ML, et al. Clinical biochemistry of domestic animals. Academic press; 2008.
- Carter SL, Rennie CD, Hamilton SJ, et al. Changes in skeletal muscle in males and females following endurance training. Can J PhysiolPharmacol. 2001;79(5):386-92.
- Dudley GA, Abraham WM, Terjung RL. Influence of exercise intensity and duration on biochemical adaptations in skeletal muscle. J Appl Physiol. 1982;53(4):844-50.
- Muylle S, Simoens P, Lauwers H, et al. Age Determination in Mini-Shetland Ponies and Donkeys. J Vet Med. 1999;46(7):421-9.
- Vall E, Ebangi AL, Abakar O. Development of a scoring system for the status of draft donkeys in the north Cameroon=Scoring to assess the status of the donkeys in north Cameroon=Realización de unaescala de Notacion del estado corporal de los asnos de traccion. Review Elev Med Vet Country too. 2001;54(3-4):255-62.
- PitelPh, Moulin M, Valette JP, et al. Approach to haematological and biochemical values in two asine breeds. Prat Vet Eq.2006;38:19-25.
- Homeida KA.Some physical variables, biochemical and haematological parameters in Hassawi ass. Scientific Journal of King Faisal University (Basic and Applied Sciences). 2005;6(1):1426.
- Mori E, Fernandes WR, Mirandola RM, et al. Reference values on serum biochemical parameters of Brazilian donkey (Equusasinus) breed. J Eq Vet Sci. 2003;23(8):358-64.
- Jordana J, Folch P, Cuenca R. Clinical biochemical parameters of the endangered Catalonian donkey breed: Normal values and the influence of sex, age, and management practices effect. Res Vet Sci. 1998;64(1):7-10.
- French JM, Patrick VH. Reference values for physiological, haematological and biochemical parameters in domestic donkeys (Equusasinus). Eq Vet Educ. 1995;7(1):33-5.
- Nininahazwe PC.Determination of a barium formula for donkeys in West Africa (Senegal, Burkina Faso, Mali and Niger). Master's thesis, EISMV, Dakar, 2016.
- Stanisic LLN, Radovic I, Ivankovic A, et al. Morphological, biochemical and hematological characterization of endangered Balkan donkey breed. ActaVeterinaria-Beograd. 2015;65(1):125-36.
- Woodroffe R, Thirgood S, Rabinowitz A. The future of coexistence: Resolving human-wildlife conflicts in a changing world. Rustica Ed. 2005;9:388.
- Evans GO, editor. Animal clinical chemistry: A practical handbook for toxicologists and biomedical researchers. CRC Press; 2009.
- Marshall WJ, Bangert SK. Clinical chemistry. 5th ed. London Elsevier. 2005.
- Girardi AM, Marques LC, Zilda C, et al. Biochemical profile of the Pêga donkey (Equusasinus) breed: Influence of age and sex. CompClinPathol. 2014;23(4):941.
- Etana KM, Jenbere TS, Bojia E, et al. Determination of reference hematological and serum-biochemical values for working donkeys of Ethiopia. Vet Res. 2011;4(3):90-94.
- Folch P, Jordana J, Cuenca R. Reference ranges and the influence of age and sex on haematological values of the endangered Catalonian donkey. Vet J. 1997;154(2):163-8.
- Caldin M, Furlanello T, Solano-Gallego L, et al. Reference ranges for haematology, biochemical profile and electrophoresis in a single herd of Ragusana donkeys from Sicily (Italy). CompClinPathol. 2005;14:5-12.
- Orlandi M, Curadi MC, Leotta R, et al. Metabolic profile in amine amine. Ann Fac Med Vet Pisa. 1997;50:47-53.
- Zinkl JG, Mae D, Guzman MP, et al. Reference ranges and the influence of age and sex on hematologic and serum biochemical values in donkeys (Equusasinus Am J Vet Res.1990;51(3):408-13.
- Sow A, Sidibe I, Kalandi M, et al. Biochemical changes induced by natural infection of trypanosomosis in Burkinabese local donkey breeds. CompClinPathol. 2014;23:103-9.