All submissions of the EM system will be redirected to Online Manuscript Submission System. Authors are requested to submit articles directly to Online Manuscript Submission System of respective journal.
Prostate-specific Antigen Journals
Prostate-specific antigen, or PSA, is a protein produced by normal, as well as malignant, cells of the prostate gland. The PSA test measures the level of PSA in a man’s blood. For this test, a blood sample is sent to a laboratory for analysis. The results are usually reported as nanograms of PSA per milliliter (ng/mL) of blood. The blood level of PSA is often elevated in men with prostate cancer, and the PSA test was originally approved by the FDA in 1986 to monitor the progression of prostate cancer in men who had already been diagnosed with the disease. In 1994, the FDA approved the use of the PSA test in conjunction with a digital rectal exam (DRE) to test asymptomatic men for prostate cancer. Men who report prostate symptoms often undergo PSA testing (along with a DRE) to help doctors determine the nature of the problem. In addition to prostate cancer, a number of benign (not cancerous) conditions can cause a man’s PSA level to rise.High Impact List of Articles
-
A Review on Nanomedicine in the Cancer Therapy
Poonam Jaggi, Harshit Joshi -
A Review on Nanomedicine in the Cancer Therapy
Poonam Jaggi, Harshit Joshi -
Cancer Metastasis and its Suppressor Genes
Raja Ramesh G.V -
Cancer Metastasis and its Suppressor Genes
Raja Ramesh G.V -
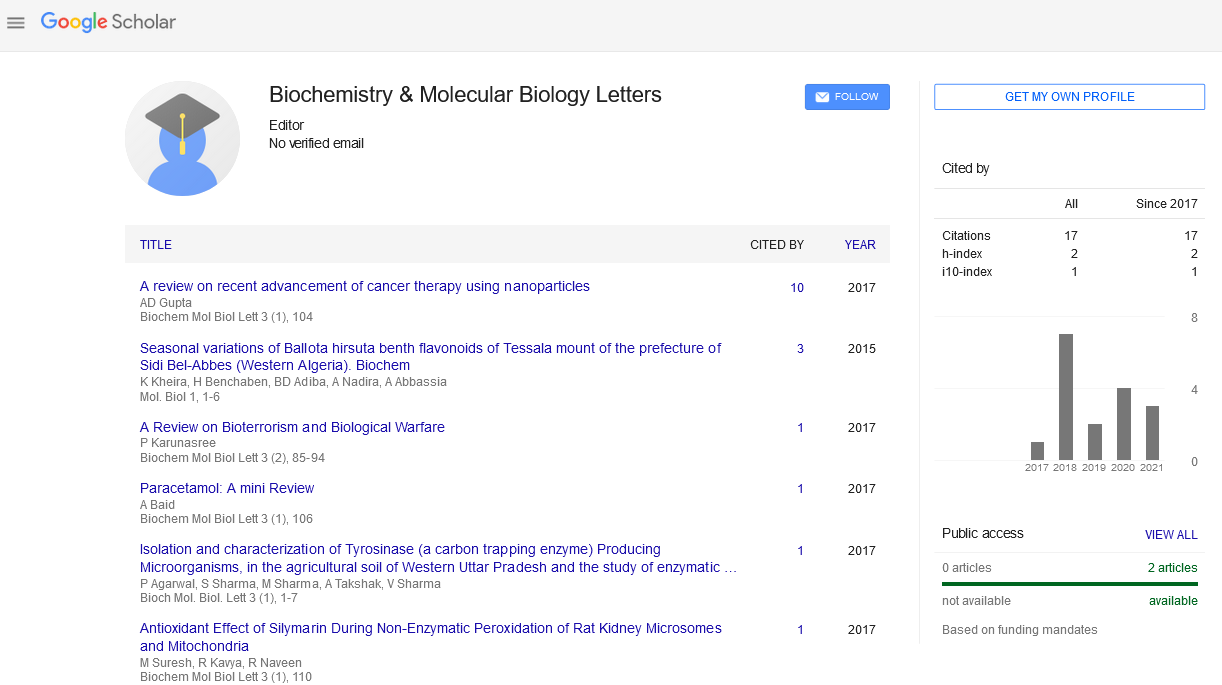
A Review on Recent Advancement of Cancer Therapy using Nanoparticles
Arka Dutta Gupta -
A Review on Recent Advancement of Cancer Therapy using Nanoparticles
Arka Dutta Gupta -
Incidence and Prevalence of Breast Cancer in USA
Anitha Sri S and Prudhvi Raj P -
Incidence and Prevalence of Breast Cancer in USA
Anitha Sri S and Prudhvi Raj P -
Advances in the Treatment of Primary Liver Cancer
PengpengOriginal Article: Biochemistry & Molecular Biology Letters
-
Advances in the Treatment of Primary Liver Cancer
PengpengOriginal Article: Biochemistry & Molecular Biology Letters